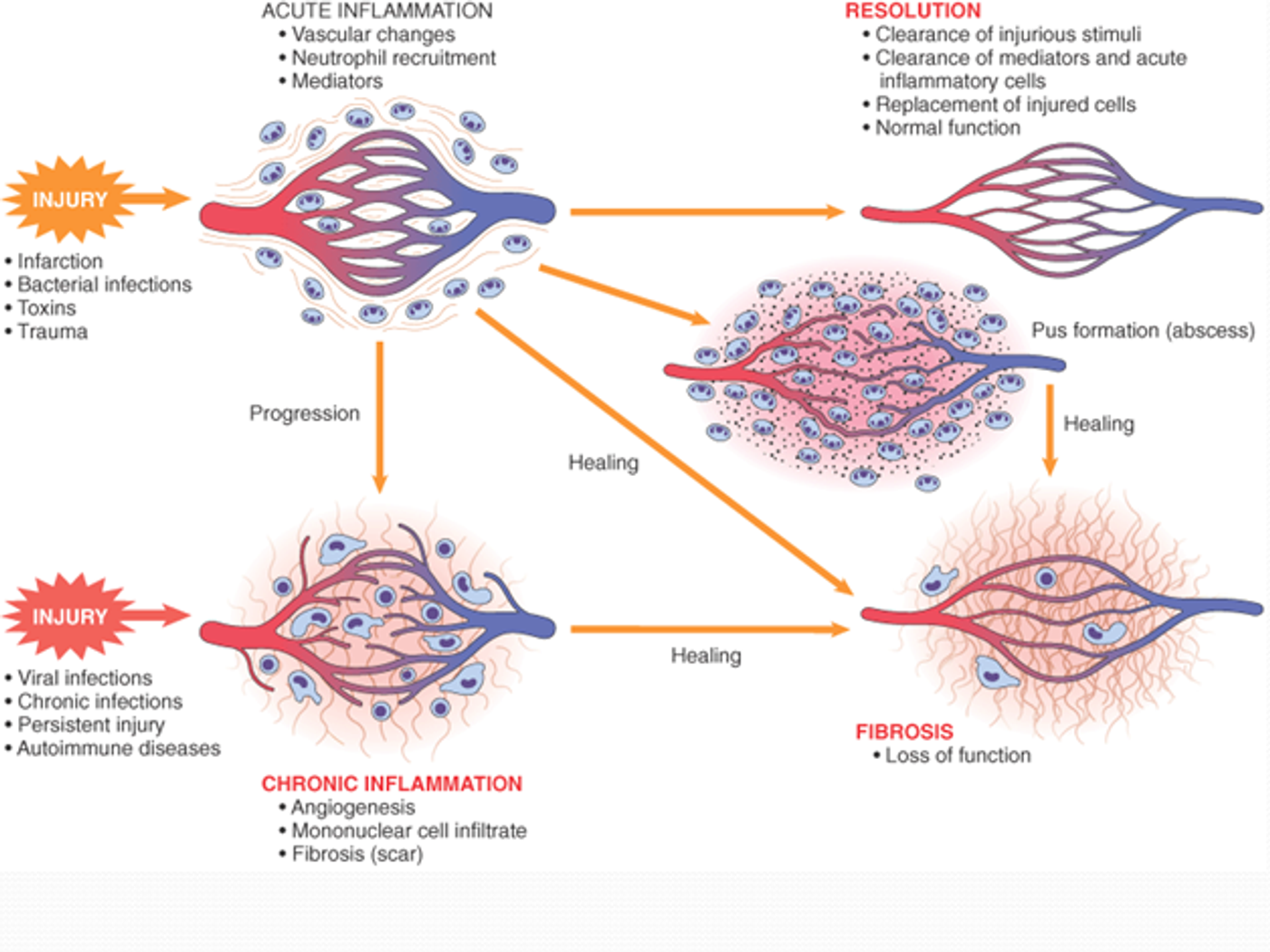
The inflammatory response is vital for fighting off infections, but too much inflammation can damage the body tissues and cause a variety of disorders. Fortunately, our immune cells have a procedure for controlling inflammation so just the right amount is administered. Scientists from Georgia State University identified a few mechanisms behind inflammation control that have potential to be used in new therapeutic options for autoimmune disorders related to inflammation, like rheumatoid arthritis.
Toll-like receptors (TLRs) are some of the first cells to respond to a foreign invader. Georgia State University researchers studied closely the interaction between TLRs and myeloid differentiation factor (MyD88) to identify how the immune system regulates inflammation.
“Understanding how the signaling of this protein is tightly regulated likely could lead to the development of novel therapeutics for many diseases,” said Dr. Jian-Dong Li from Georgia State University. His team’s research was published in the journal Proceedings of the National Academy of Sciences.
Their study looked to identify specifically how MyD88-mediated signaling is controlled, hoping to gain insight into the natural ways the body maintains inflammatory homeostasis. When a pathogen is detected, MyD88 “functions as an essential signal transductor” for the TLR and interleukin pathways (
NCBI Gene ID). Without MyD88, the body would be exceedingly vulnerable to certain infections.
The Georgia State University team also identified the role of CYLD protein key player in controlling MyD88 activity. This protein blocks MyD88 signaling at lysine 231, a critical amino acid for transduction. Without CYLD, there would be no maintenance of the immune response. Without a balance, the body tissues could be damaged from unnecessary inflammation.
By stimulating an immune response in mice and human epithelial cells with nontypeable Haemophilus influenzae (NTHi), a virus responsible for chronic obstructive pulmonary disease (COPD) and ear infections, the research team could directly observe the interaction between CYLD and MyD88.
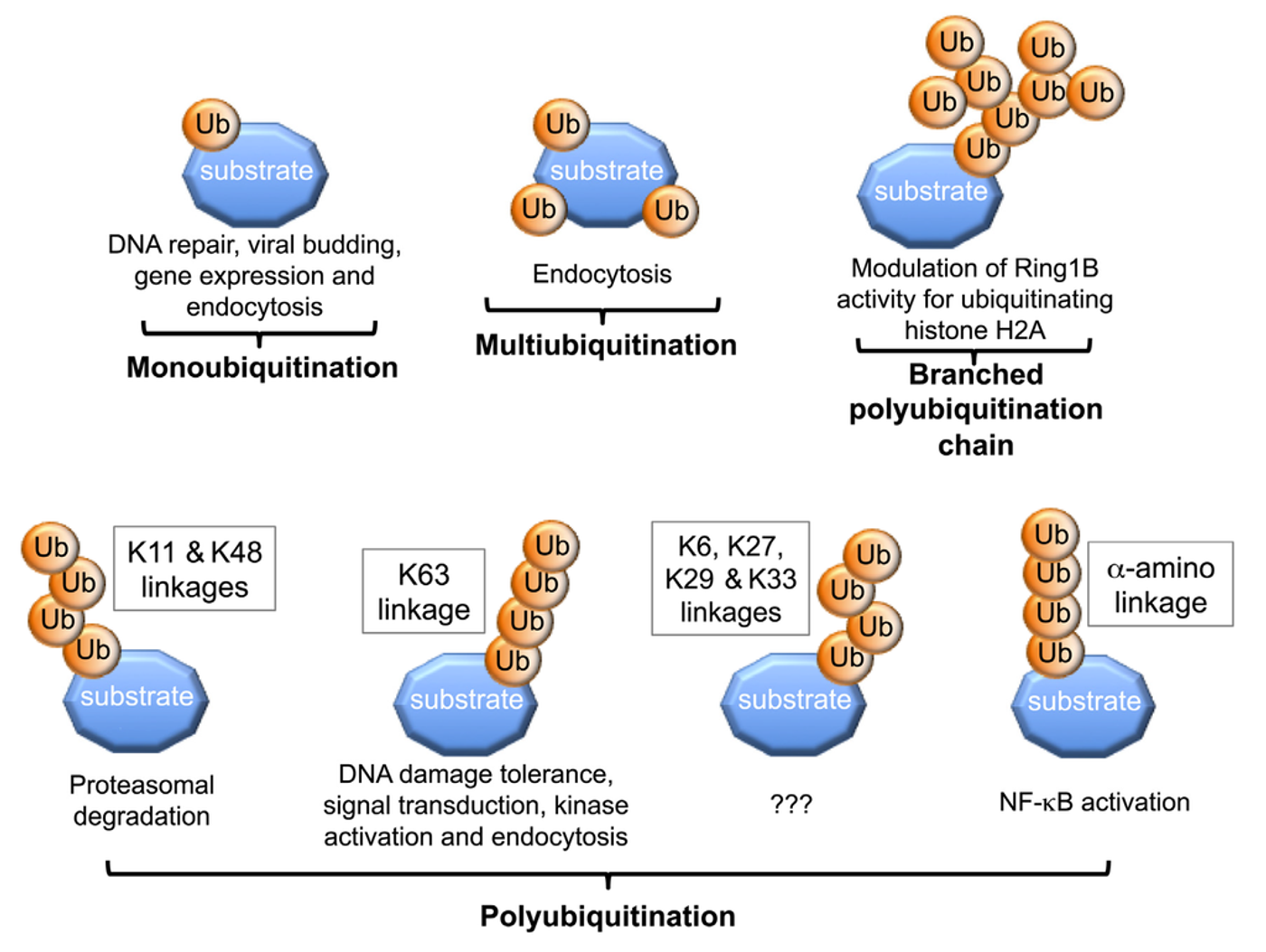
They found that NTHi prompts polyubiquitination of MyD88, a process which facilitates TLR signaling from MyD88 proteins. Polyubiquitination occurs when ubiquitin protein attaches to lysine residues (
Cell Division).
Next, they found that CYLD controls inflammation through inhibition of MyD88 polyubiquitination by either deactivating the bacteria or reversing the polyubiquitination. CYLD is a protein coding gene that “functions as a deubiquitinating enzyme” (
GeneCards).
“Because we know this is a key therapeutic target, we can develop therapeutics specifically against this target in the future--for instance, treatments that increase the production of the brake-pedal protein CYLD,” said Li.
Watch this video to learn about how inflammation impacts the body: