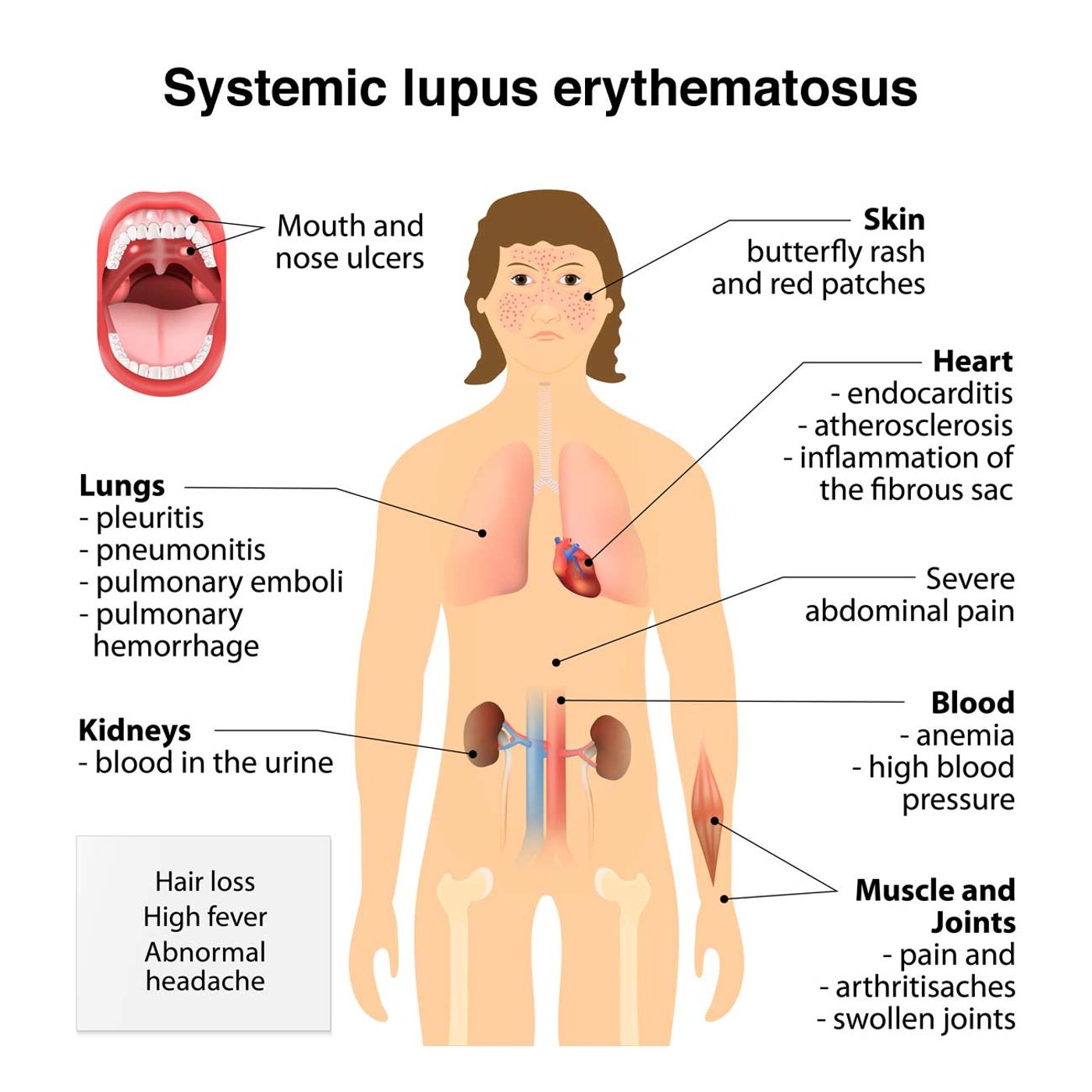
Systemic lupus erythematosus (SLE) is a complex autoimmune disease that revolves around the immune system’s error as it attacks healthy host cells. A domino-effect type of pathway leads to the unnecessary inflammation that characterizes this disease, and long-time SLE investigators from the Baylor Institute for Immunology Research are inching closer to a new treatment for this disease.
When SLE patients’ immune systems mistakenly attack healthy tissues, the skin, joints, kidneys, and brain can all be targeted (
MedlinePlus). While there is no current cure for this condition, doctors usually focus on controlling symptoms with various drugs once people receive their diagnosis:
- Nonsteroidal anti-inflammatory drugs (NSAIDs)
- Corticosteroids
- Immunosuppressants
A previous study from Baylor outlined the pathway of pathogenesis that produces SLE symptoms, although the ultimate and initial trigger causing SLE is still unknown. Immune cells called neutrophils err by responding to autoantibodies, and then they force the autoantibodies to release some DNA. Next, another immune cell called plasmacytoid dendritic cells (pDCs) are activated, which in turn prompt type I interferon to activate the immune system.
During a healthy and normal immune response, pDCs respond to foreign nucleic acids rather than host nucleic acids (
Annual Review of Immunology). These dendritic cells also have antigen-presenting capabilities that make them powerful players in both the innate and adaptive immune responses.
In a new Baylor study published in
The Journal of Experimental Medicine, scientists looked even closer at the SLE immune pathway to see if there was any conceivable way to interrupt it in order to develop new ways to treat both SLE and other chronic autoimmune diseases.
"Therapeutic efforts to enhance pathways involved in oxidized mitochondrial DNA degradation should be explored in human SLE, a disease for which only one new drug has been approved in the past 50 years," said Baylor scientist Virginia Pascual, PhD.
New findings from the current paper showed that oxidized DNA accumulates inside of the mitochondria of neutrophils, eventually extruded to “potently” stimulate plasmacytoid dendritic cell production of type I interferons, which they already knew would result in stimulating of the immune response.
Mitochondria usually degrade oxidized DNA via lysosomes rather than releasing the genetic material into the cell. They do so by disassembling their nucleoids, the intracellular structures that contain packaged DNA, and transfer the oxidized DNA to the lysosomes. However, the scientists from the current study saw that SLE patients’ mitochondrial process of oxidized DNA degradation is interrupted with neutrophils recognize autoantibodies that are present. This event interferes with nucleoid disassembly, the oxidized DNA is kept inside the mitochondria for a dangerously extended time, and then it is released from the cell instead of being degraded, which is where dendritic cells and type I interferon stimulate the immune system.
Baylor researchers are hopeful that the information gleaned from this study will provide strong support for developing new treatments for SLE, such as targeting oxidized DNA.
Source:
Baylor Institute for Immunology Research