Scientists learn more everyday about the complex and unique population of microbes in the human gastrointestinal (GI) tract. While these “good bacteria” serve a variety of beneficial functions for the gut like helping facilitate digestion, any dislocation of these bacteria into the bloodstream could cause infection elsewhere in the body that isn’t equipped to peacefully coexist with the microbiota. To prevent systemic infection from occurring, there is a thin layer of cells that provides a barrier between the microbiota and the rest of the body. A new study from The Rockefeller University investigates the content of this layer and how the body maintains a healthy population of gut bacteria without infecting the rest of the body.
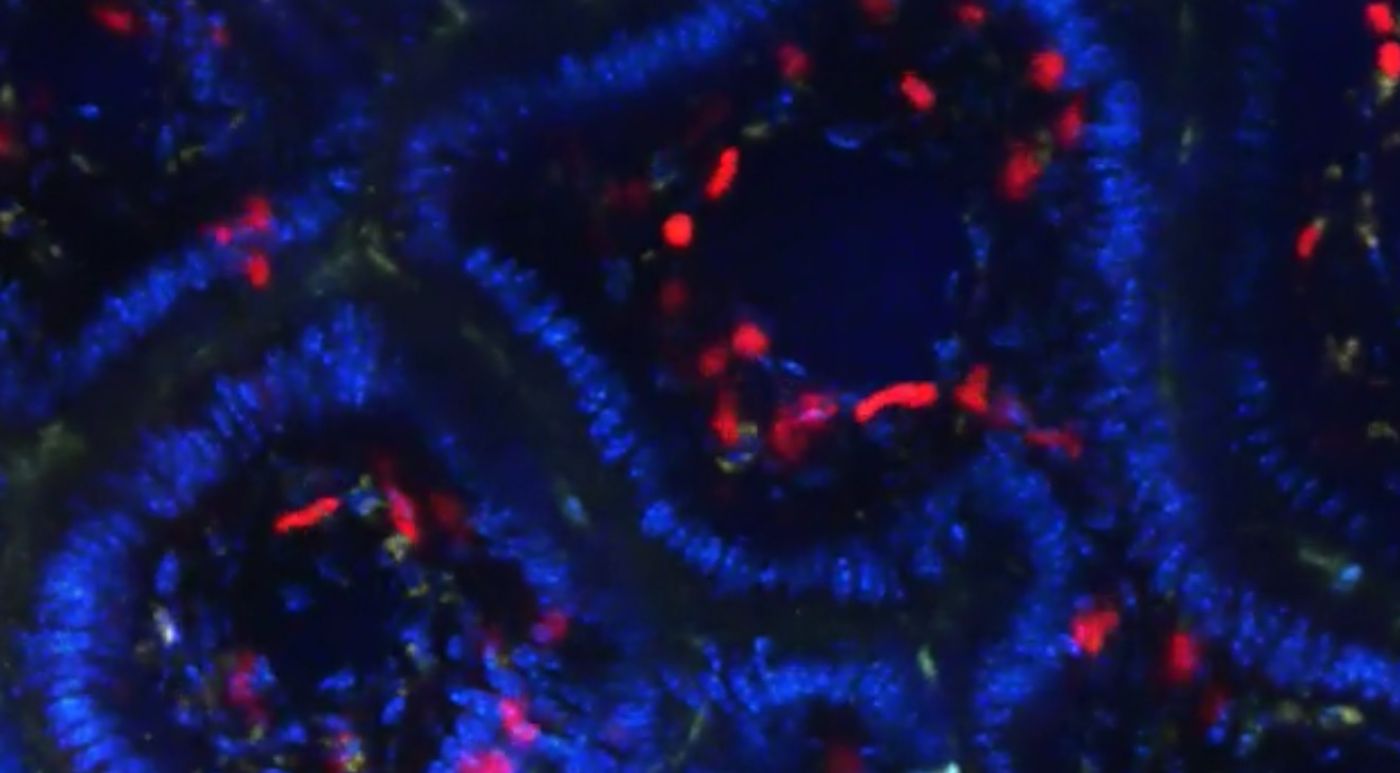
You fall and scrape your knee, or you catch a nasty cold from someone on the bus. Instantly, your immune system is responding to the damage, with the first response almost always being inflammation: a rush of blood, carrying lymphocytes, to the site of injury or infection. There is also a component of the immune system that serves to make sure inflammatory cells don’t get carried away: regulatory T cells.
After examining the barrier layer of the GI tract, scientists found a surprising lack of regulatory T cells (Tregs), the lymphocyte population that is known for mediating the immune response throughout the body. Instead, they found intraepithelial (IEL) CD4 cells. However, they weren’t so sure that Tregs still weren’t involved, and further studies indeed showed a connection between IEL CD4 cells and Tregs.
IEL cells are also found in other epithelial cells of the body: skin, biliary tract, oral cavity, upper respiratory tract, lungs, and the reproductive tract. These lymphocytes are some of the most diverse of the body’s lymphocyte populations.
Researchers from the current study, published in
Science, discovered that the IEL CD4 cells they found in the barrier layer of the GI tract were descendants of Tregs that had first populated the gut lining. To figure out how the lymphocytes made the transition into IEL CD4 cells, the team started tracking Tregs as they made the trip to the GI tract.
Over five weeks, they saw half of the labeled Tregs that traveled to the gut lining eventually stopped expressing a protein called Foxp3. The gene that encodes this protein is a transcriptional regulator whose defects have been related to disorders like immunodeficiency polyendocrinopathy, enteropathy, and X-linked syndrome. Of the Tregs not expressing Foxp3, a fraction transformed into IEL CD4 cells, making the Rockefeller scientists the first to record the transformation of Tregs into another cell type in a living organism.
This discovery also provides a generous piece of the puzzle for scientists who study the diversity of the microbiome and how reducing certain populations can be harmful to human health. Mice studies from the same researchers show a response from the gut bacteria to the Treg conversion to IEL CD4 cells which seems to have a role in the conversion process. However, with certain antibiotic treatments, the Tregs stopped converting to IEL CD4 cells. How common this response is in human life and how it effects the health of the microbiome, researchers are still unsure.
Sources:
The Rockefeller University,
Gene Cards Human Gene Database,
Encyclopedia of Life Sciences