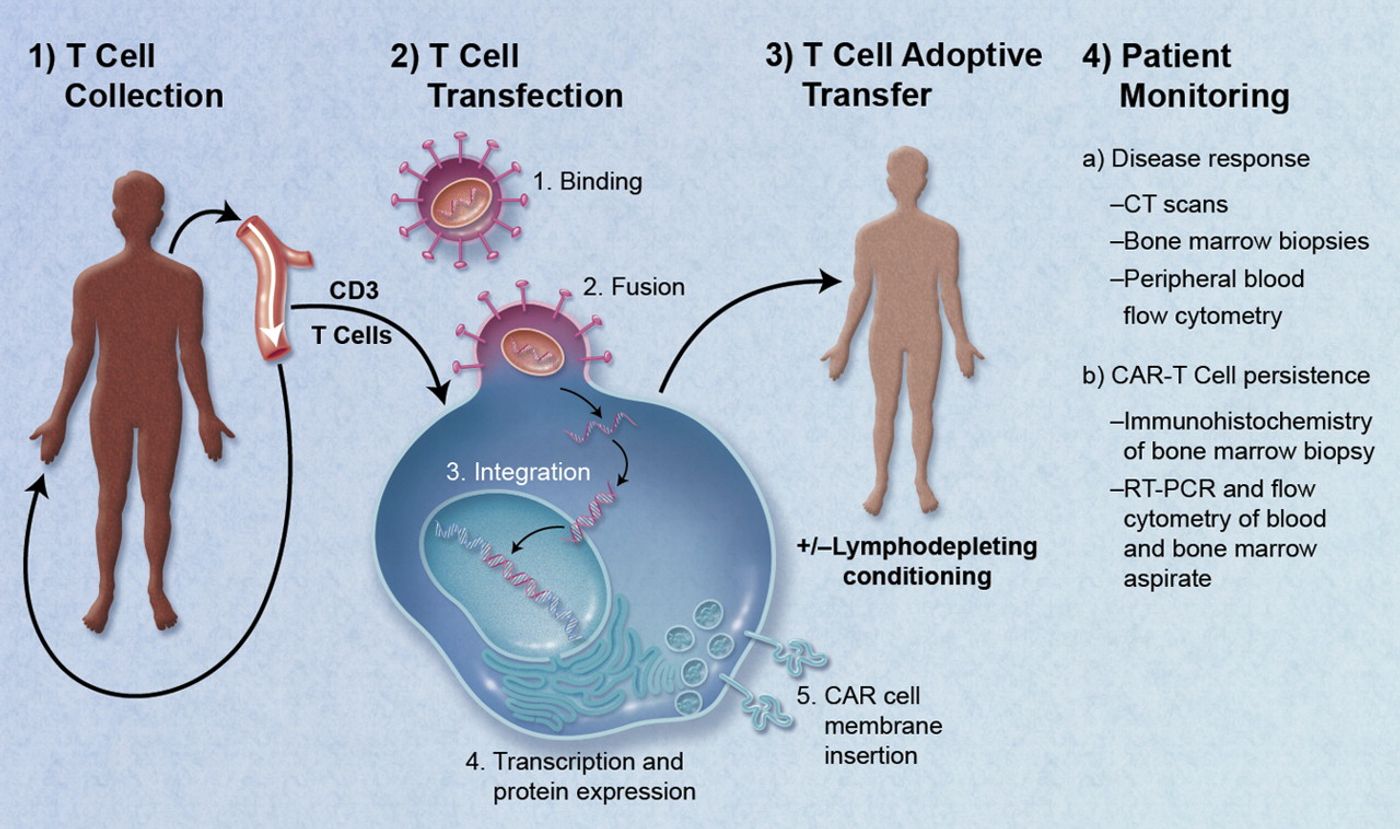
T cells are the body’s natural fleet of cancer-attacking lymphocytes, but in recent years scientists have found a way to engineer their own version to be even more effective at targeting tumors. The engineered cells, called chimeric antigen receptor (CAR) T cells, were first developed in the 1990s, and scientists have been making improvements ever since. In a new study published in
Immunity, scientists discuss their latest modification: enabling CAR T cells to attack solid tumors in addition to blood cancers.
Scientists design these “bigger and better” cells first by extracting a patient’s own T cells. Then, the lymphocytes are equipped with cancer-specific surface proteins to help them better recognize harmful, growing tumors. Then, the patient gets the T cells back, more armed and ready to attack than ever before.
In the past, researchers had trouble with CAR T cells responding too strongly to cells in the body – killing healthy cells in addition to tumor cells. Scientist introduced a regulatory process
in a recent study from Purdue University to ensure the CAR T cells would have the right cancer-attacking abilities without committing any friendly fire. Similarly, in a new study from the University of Pennsylvania Perelman School of Medicine, researchers successfully identified a way to enable CAR T cells to target solid tumors specifically, leaving surrounding healthy tissue alone.
The secret to their success was equipping T cells with a combination of cancer-specific proteins: a known cancer-associated surface protein and a cancer-associated carbohydrate. Healthy cells have an immature version of the surface protein inside of them, but the CAR T cells only recognize the protein when it is on the surface of the cell. It just so happens that cancer cells bring the immature version of the protein from inside the cell to the surface, making them visible and vulnerable to CAR T cells on the prowl.
The team from Pennsylvania studied mouse models of leukemia and pancreatic cancer to test the ability of the newly-engineered CAR T cells. Their version of CAR T cells expressed an antibody that bound specifically to truncated carbohydrate molecules on a protein called mucin 1 (MUC1). Because proteins like MUC1 exist inside of the cell unless brought to the surface by cancer, CAR T cells specifically engineered to target MUC1 can lead to cancer drugs that reduce toxicity.
After treating the mice with injections of CAR T cells, the researchers saw a widespread reduction of tumor volume, and most of the mice had their tumors eliminated completely. After 113 days, mice with pancreatic cancer treated with the experimental CAR T cells were still alive. Unfortunately for this mice with CAR T cells that were not programmed to target MUC1, only one-third of the population survived. The researchers saw no damage from CAR T cells to healthy cells without MUC1 on their surface, further confirming their non-toxic quality.
“We are continuing to evaluate the safety of this therapy in additional models,” said co-senior author Laura Johnson, PhD. “Once the new models are complete, we plan to move this therapy into a phase I clinical trial for patients with solid tumors.”
Johnson and her team can move forward with confidence; the newly-designed CAR T cells will not attack healthy cells because they can’t. Their specific design is the key to providing a non-toxic option for future immunotherapies, as well as provide a treatment specific to certain types and sub-types of cancer. Researchers look forward to welcoming this new, safe, and effective treatment for cancer patients.
Sources:
University of Pennsylvania School of Medicine,
National Cancer Institute