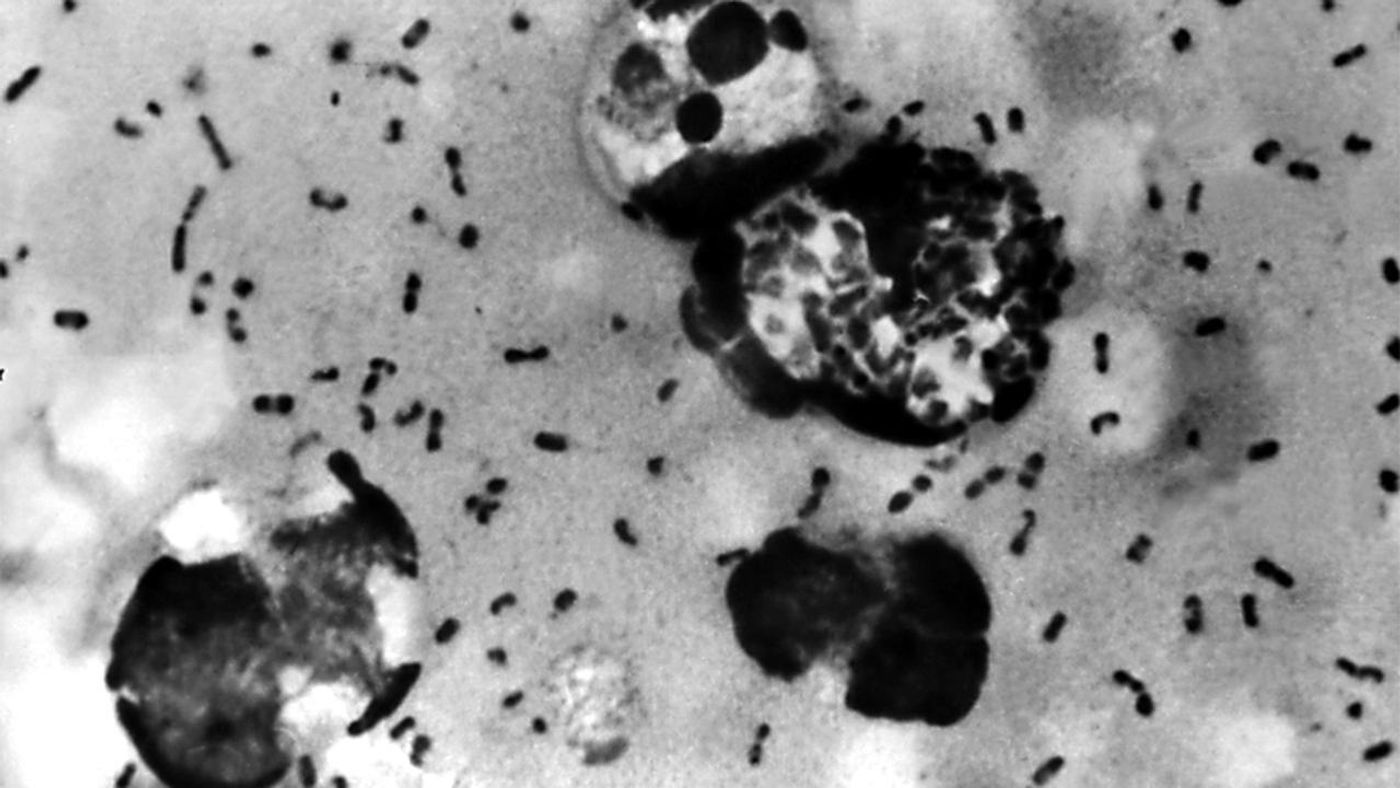
Yersinia pestis, the bacterial species responsible for numerous and deadly outbreaks of plague throughout human history, contains a toxin that might actually be beneficial to human health.
The same bacterial toxin that caused the 15th century “Black Death” epidemic of bubonic plague by invading host cells and disrupting valuable signaling responses to promote pathogenesis could be used to treat a common inherited autoimmune disease called Familial Mediterranean Fever (FMF). The autoinflammatory condition affects 1 in 200 to 1 in 1000 people in populations originating from the Mediterranean region, namely those with Armenian, Arab, Turkish, or Jewish ancestry.
Individuals with FMF have recurring episodes of painful inflammation in the abdomen, chest, or joints often accompanied by fever, rash, and headache, with initial episodes usually occurring during childhood or the teenage years. Episodes can be as short as 12 hours and as long as three days.
Lack of proper intervention in patients with FMF can lead to organ failure due to amyloidosis, the buildup of protein deposits in the body’s organs and tissues. FMF is passed down through generations in an autosomal dominant pattern, meaning it’s possible for a child to inherit the disease from just one affected parent.
The symptoms of FMF stem from mutations in a gene called MEFV, which is responsible for making a protein called pyrin, found in leukocytes. Pyrin is a regulator of inflammation, and when MEFV mutations inhibit ample production of pyrin, the inflammatory process can grow largely out of control.
Lead author of the recent
Cell Host & Microbe study James Bliska, PhD., said the findings “may explain the natural selection process behind a chronic condition that affects a high prevalence of people originating around the Mediterranean Sea.” Bliska is a Professor in the Department of Molecular Genetics & Microbiology at Stony Brook University School of Medicine.
Bliska and his team researched the Y. pestis toxin, also found in bacterial species Y. pseudotuberculosis, because of its ability to recruit human kinases PRK1 and PRK2 to phosphorylate pyrin and subsequently restrict its inflammasome from causing an over-the-top autoimmune response from FMF-related MEFV mutations.
So-called Yersinia outer proteins (Yops) promote pathogenesis by disrupting host signaling responses that control the immune response to bacterial infections. Miraculously, the same disruption that caused multiple epidemics and taking hundreds of thousands of lives could also be able to treat a common autoimmune disease with no known cure.
Bliska and his team will continue to unravel the relationship between Yops and the control of FMF, encouraged by the increasing potential for drug developers to create new therapies that mimic this method of immune inhibition.
Sources:
Stony Brook University,
Blood Journal,
Genetics Home Reference