A new drug candidate for multiple sclerosis (MS) was successfully tested in mice models of the disease. The drug, called laquinimod, carefully prevents the entrance of autoimmune cells from entering the central nervous system (CNS), without inhibiting body-wide immunity.
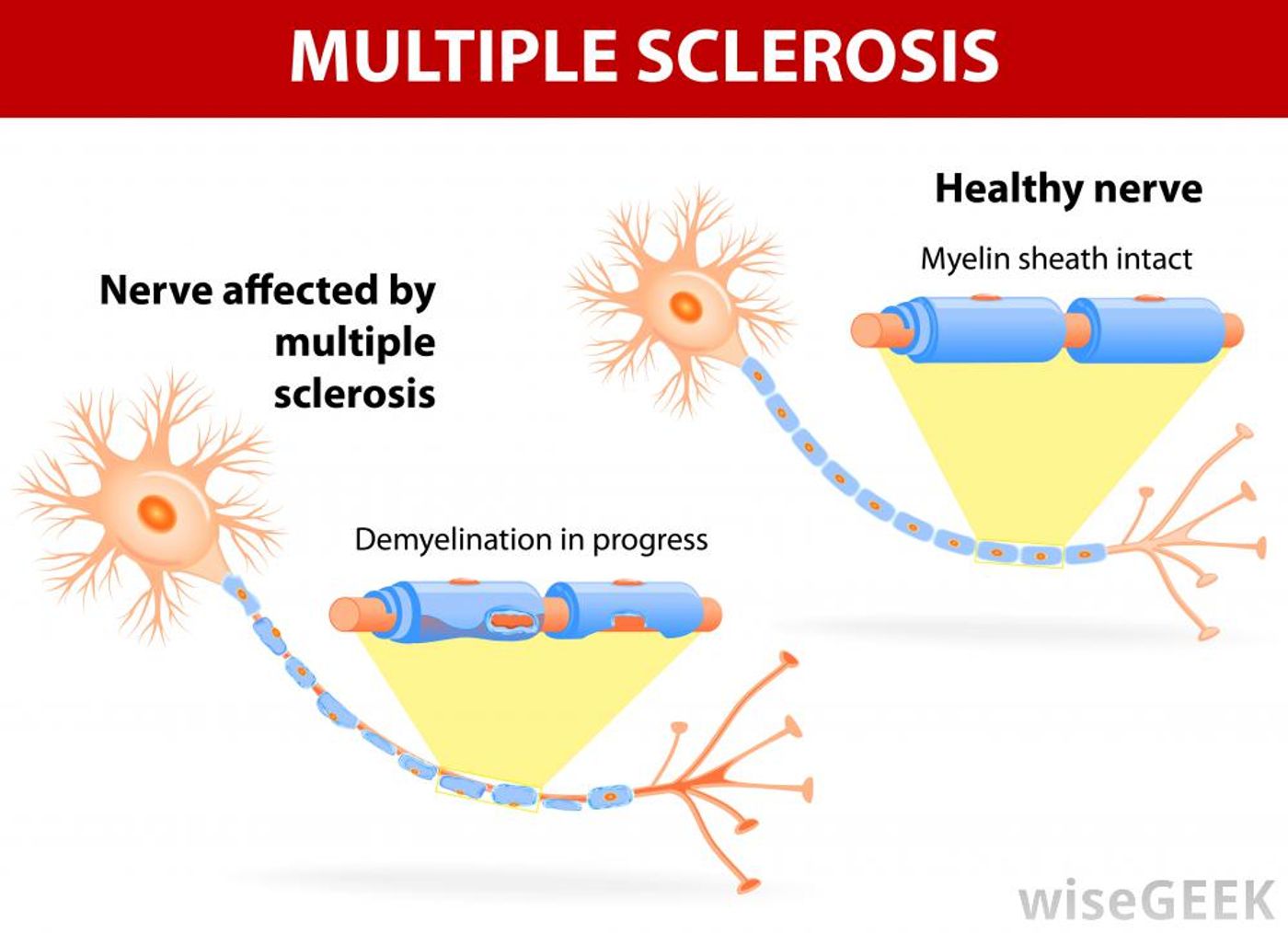
In individuals with the autoimmune disorder MS, lymphocytes called T cells and B cells work together to produce antibodies that attack and destroy the nerve-insulator, the myelin sheath. Without myelin, the CNS nerves are massively damaged, leading to a slew of disease characteristics such as weakness, numbness, loss of muscle coordination, and problems with vision, speech, bladder control. However, the production of antibodies through T cell and B cell teamwork is also what protects the body from infection, and scientists have been trying to understand what makes these cells “go rogue” in autoimmune diseases like MS.
Regardless of why the immune cells act like they do in cases of MS, the experimental drug laquinimod showed promise in its ability to prevent the progression of the disease in a new study from the American Academy of Neurology, recently published in the journal
Neurology Neuroimmunology & Neuroinflammation.
Scientists used a mice model of MS called spontaneous experimental autoimmune encephalomyelitis (EAE), a commonly-used model system for studying MS in the lab. Researchers send T cells into the CNS pre-programmed to target myelin, mimicking the effects that occur in natural cases of MS. Although myelin is present to protect nerves in both the central nervous system (CNS) and the peripheral nervous system (PNS), only the CNS is affected in MS.
In the first of two studies, researchers compared application of a daily oral dose of laquinimod versus a placebo in mice models of EAE. After collecting T cell and B cell counts, they could make observations as to how the drug was affecting the progression of the disease. Out of 50 mice from the initial study, less than thirty percent of mice receiving laquinimod developed MS, and nearly sixty percent of the mice given placebo did develop the disease.
The first study also showed a nearly 100 percent reduction in meningeal B cell aggregates in the mice receiving the experimental drug, and these types of aggregates are only found in cases of progressive MS.
Next, they gave laquinimod to 22 mice with MS only after the mice had already developed paralysis from EAE. However, administration of the drug resulted in a visible reduction of the progression of the disease. Additionally, compared to control groups who did not receive the drug, the 22 experimental mice showed a 49 percent reduction in dendritic cells, a 46 percent reduction in T follicular helper cells, and a 60 percent reduction in antibodies targeting myelin. Dendritic cells promote the production of T follicular helper cells, which are essential for B cells to create autoantibodies in MS.
Overall, laquinimod successfully proved its ability to treat MS-related lymphocyte migration to the CNS in several ways. The drug’s specificity prevents other aspects of the otherwise-healthy immune system from being damaged, and the study authors believe the drug to be capable of providing “insight regarding the potential application of laquinimod to MS treatment.”
Sources:
American Academy of Neurology,
Multiple Sclerosis Discovery Forum,
Multiple Sclerosis Society,
PubMed Health Glossary,
Immunity