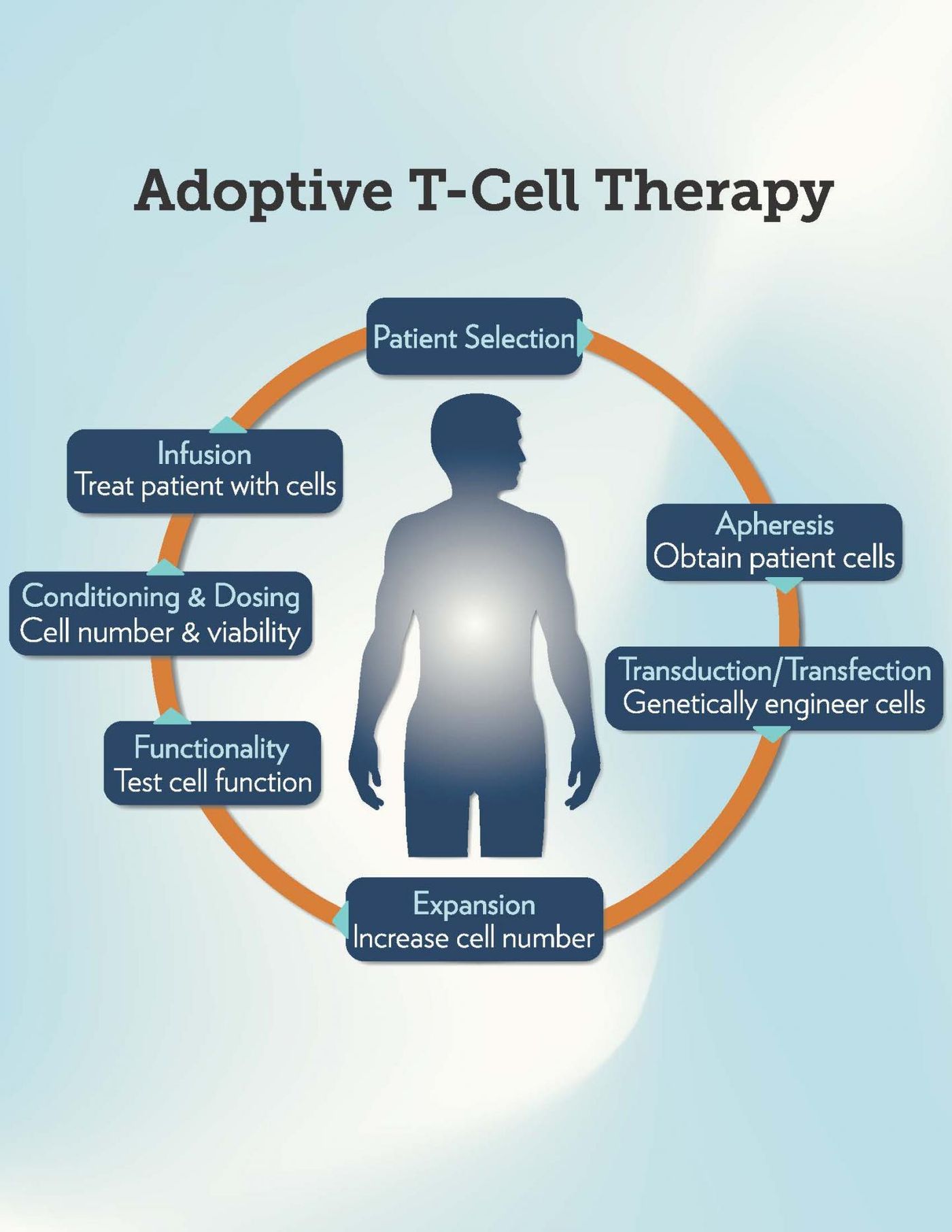
Enhancing a cancer patient’s own T cells to reduce tumor growth is an up-and-coming cancer therapy that is being used to treat multiple types of tumor growth. A new study from the Medical University of South Carolina, in collaboration with Loyola University, offers a new way to insure that the enhanced T cells survived once they re-enter the body.
Adoptive cell therapy (ACT) began as an immune-boosting cancer treatment as scientists began infusing autologous, melanoma-specific T cells obtained from the patient’s own supply of cells. After obtaining the cells, researchers genetically modify them with therapeutic receptors, activate them, and then stimulate them to proliferating before facilitating a therapeutic re-infusion back into the patient’s bloodstream.
Unfortunately, rapid proliferation before reinfusion makes T cells increasingly prone to activation induced cell death (AICD), and the best results only occur when the transferred enhanced T cells survive. Fast-forward to this study and the authors’ current goals: how can they prevent AICD to enhance ACT’s effectiveness at targeting cancer, mainly melanoma? After extensive experimentation, they found that culturing the T cells in N-acetyl cysteine (NAC) before re-infusion protects them from AICD, making the immunotherapy increasingly effective and outcomes much more promising in a preclinical model of melanoma.
The collaborative effort led to the researchers seeing significant improvements in T cell persistence and overall outcomes when they added NAC to the in vitro T cell expansion culture. Apparently, NAC prevents increases in a DNA damage marker called yH2AX. Specifically, 40 percent of NAC-cultured T cells were detectable in tumors post-re-infusion as compared to only 1.2 percent of standard-culture T cells.
"We were really surprised," said senior author of the recent
Cancer Research study Christina Voelkel-Johnson, PhD, from the Medical University of South Carolina. "Given the harsh environment T cells encounter within tumors, we did not expect that the number of NAC-cultured T cells would be 33-fold higher than T cells not cultured in NAC." Plus, in mice models, they saw a significant delay of tumor growth in mice receiving T cell re-infusion after being cultured in NAC.
Their studies show that NAC culture reduces the level of reactive oxygen species (ROS), which reduces the amount of damage done to the DNA through an extensive molecular pathway of kinases, tumor proteins, and antibodies. When less DNA damage occurs in T cells, the body’s regulatory cells don’t have to make a choice between repair and cell death, reducing the overall rate of T cells destroyed because of DNA damage.
Next up: how does NAC culturing of T cells affect their phenotype? How does it work? In Voelkel-Johnson’s words: “There may be benefits to culturing T cells in NAC aside from generating AICD resistance that we haven't yet recognized."
More about melanoma:
Melanoma is a cancer that begins in the melanocytes. It’s less common than basal cell and squamous cell skin cancers, but it’s more dangerous because it is more likely to metastasize if not treated early. The American Cancer Society estimates that more than ten thousand people in the United States will die from melanoma in 2016.
Sources:
Medical University of South Carolina,
American Cancer Society