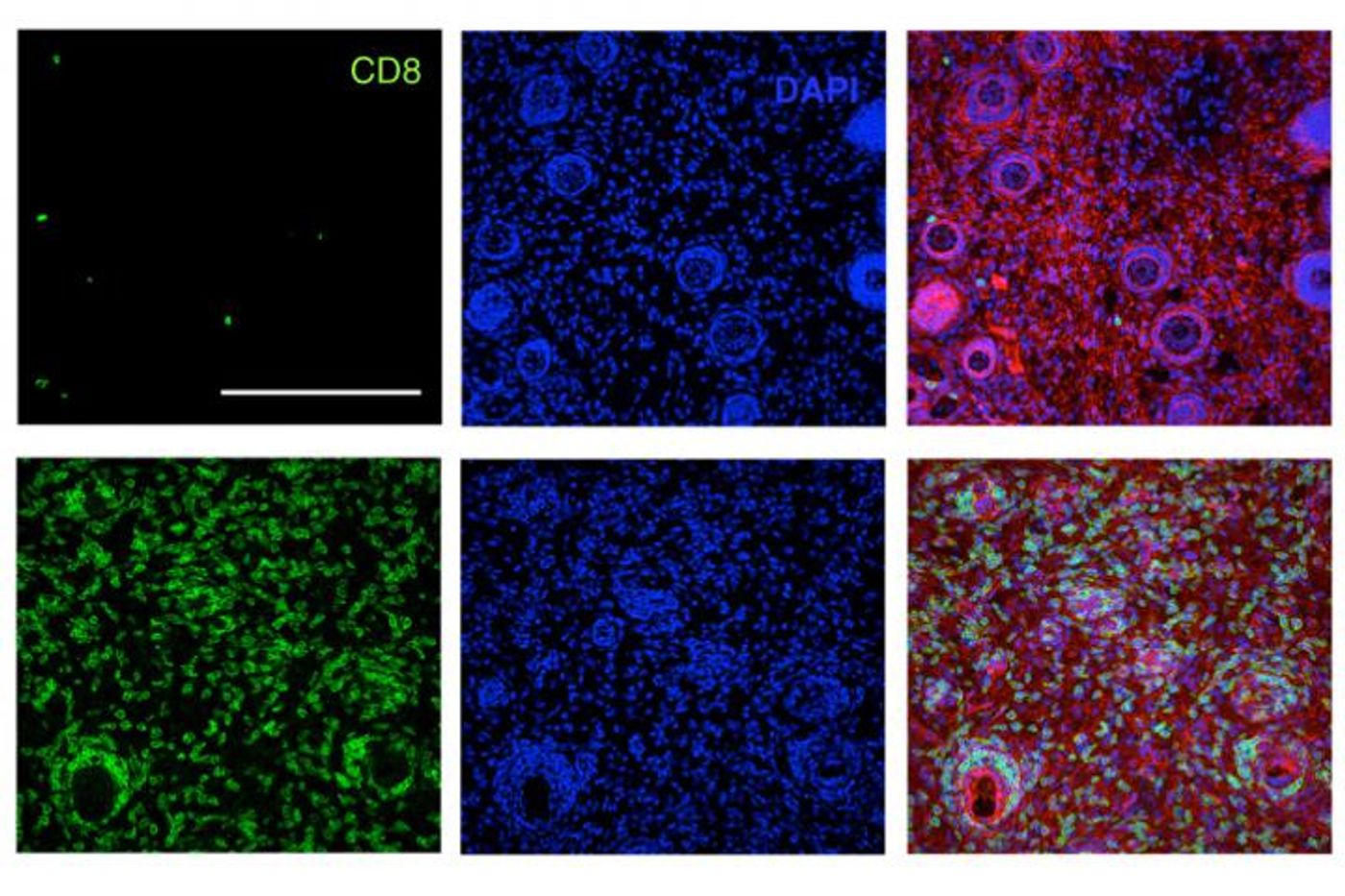
The newest effort in cancer treatment research involves the combination of not one, not two, not three, but four different cancer immunotherapies to kill tumor cells. So far in mice studies with extremely aggressive cancer, the combination treatment has been 75 percent effective.
Two scientists from the Massachusetts Institute of Technology (MIT) have brought their research together to make a cancer therapy juggernaut. First, MIT’s Dane Wittrup, PhD, studied the deliverance of antibodies and a signaling molecule called interleukin-2 (IL-2) that enhances the immune response in mice. The treatment stopped the growth of aggressive melanoma tumors in mice for as long as they received treatment. Plus, Wittrup’s findings appeared to be even more successfully when the researchers added T cells to be delivered along with antibodies and IL-2, stimulating the adaptive immune system.
Then, MIT’s Darrell Irvine, PhD, studied a novel T cell “vaccine” that attaches to lymph nodes via a protein called albumin. Once linked to the lymph nodes, the T cell vaccine stimulates large production of T cells to gang up against the tumor cells.
Once Irvine and Wittrup combined their experimental treatments, they quickly saw both powerhouses of the immune system become activated, the adaptive and the innate responses. Their findings showed that the new therapy has the potential to treat multiple types of human cancer, and it is also possible that the treatment could trigger immune memory so lymphocytes could “remember” the right cancer cells to target and kill if the tumor continues to grow.
"We wondered if we could bring these two [therapies] together and try to generate a more integrated immune response that would bring together all arms of the immune system against the tumor,” Irvine said.
The result of combining the two therapies was a four-part anti-cancer response:
- Antibody targeted tumor and recruited additional immune cells to help activate T cells
- Vaccine targeted tumor and stimulated the proliferation of T cells to attack the tumor
- IL-2 helped the T cell population expand quickly
- Anti-PD1 molecule blocked PD1, a receptor found on T cells, so the lymphocytes would stay active longer
They tested the combination treatment in mice implanted with three different types of tumors: melanoma, lymphoma, and breast cancer. These engineered cancers are harder to treat than human tumors implanted in mice because they suppress the immune system.
Overall, no matter what time of tumor the mice were implanted with, the researchers saw a general trend of tumor elimination: just about 75 percent of the tumors were completely destroyed. To confirm the staying power of the treatment, the researchers implanted more tumor cells into the same mice that had cleared their cancer. Their immune systems were once again able to eliminate all of the tumor cells.
"We have shown that with the right combination of signals, the endogenous immune system can routinely overcome large immunosuppressive tumors, which was an unanswered question," said Irvine.
While cancer treatments in the past were often thwarted by the ability of tumor cells to secrete chemicals to remain invisible to the immune system, scientists are hoping to get the immune cells back online with the new combination treatment from MIT.
"To our knowledge, nobody has been able to take tumors that big and cure them with a therapy consisting entirely of injecting biomolecular drugs instead of transplanting T cells," Wittrup said.
In the future, Wittrup, Irvine, and others from MIT plan on designing the combination treatment to treat both specific cases of cancer and broad cases of cancerous growth. Their current study was published in the journal
Nature Medicine.
Source:
Massachusetts Institute of Technology