TGF-Beta Targeted by New Antibody-Based Therapy
TGF-beta may be an “old story” in the study of cancer, but a plot twist was recently encountered by scientists from the Medical University of South Carolina. TGF-beta, which is hijacked by cancer cells to grow and avoid the immune system, needs a molecule called GARP in order to serve the functions desired by cancer cells.
Scientists have studied TGF-beta and its connection to cancer since the 1980s. TGF-beta is a cytokine employed by the immune system, mainly regulatory T cells (Tregs) to signal to other immune cells not to attack healthy, self cells. TGF-beta also regulates the cell cycle. As helpful as these two main functions are, it also makes TGF-beta a pretty clear target for tumor cells in pursuit of a cellular guide to help them evade the immune system and manipulate the cell cycle so they can grow uncontrollably.
In the past scientists have shown the massive release of TGF-beta by malignant tumors that easily divide and hide from the immune system by promoting Treg suppression of the immune response. It is difficult to develop cancer treatments that target cancer-manipulated TGF-beta because healthy, necessary forms of the cytokine need to exist elsewhere in the body.
“The new spin is that there is a docking receptor for TGF-beta that increases the activity of the cytokine, and this molecule is called GARP,” explained principal investigator Zihai Li, MD, PhD. GARP is a receptor, and the only one scientists have found to “dock” TGF-beta to the surface of cells, and this attachment is thought to help these cells store up stocks of TGF-beta.
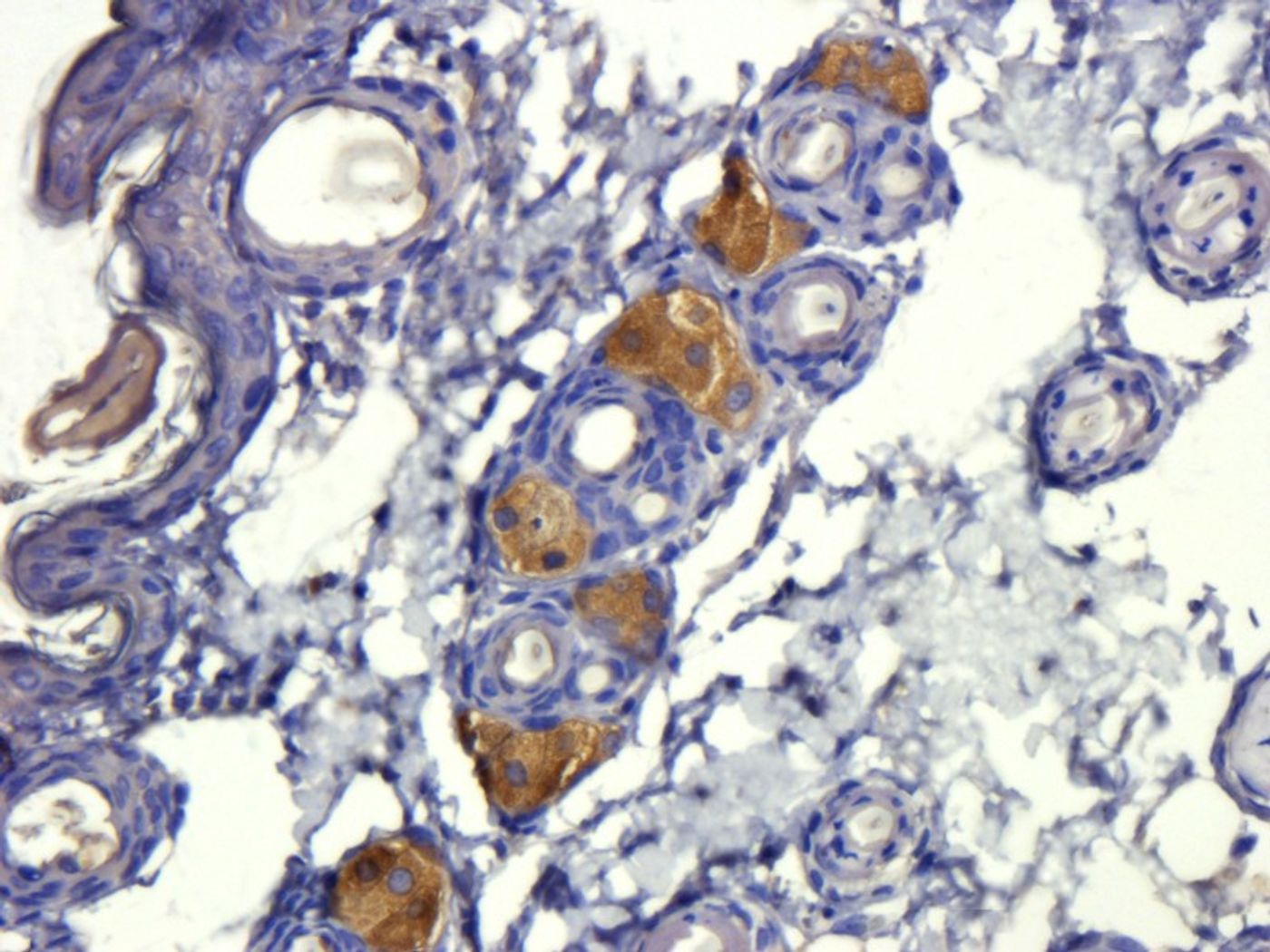
A graduate student in Li’s lab saw that levels of GARP were abnormally high in biopsies of human breast, lung, and colon tumors compared to normal tissue. This lead Li and his team to further investigate how GARP might be enabling TGF-beta to become “out of control.” The theory? More GARP in the tissues meant more storage capacity for cancer cells to hold TGF-beta for their evasion and metastasis agenda.
The researchers experimented with the deletion of the GARP gene in mice with mammary tumors. Without GARP, the tumors grew more slowly and were less able to metastasize to the lungs. But they saw increased TGF-beta signaling, tumor growth, and metastasis after the GARP gene was inserted into mouse mammary cells expressing high levels of GARP.
Additionally, mice observed to have more GARP also showed more TGF-beta-releasing Tregs. What does this means? That GARP promotes both metastasis and immune suppression in cases of breast cancer.
Li’s findings were not all doom and gloom, fortunately. The researchers also tested out a series of antibodies with which they immunized the mice models. One antibody, called 4D3 was successful in directly blocking human TGF-beta from binding to GARP expressed on cell surfaces. 4D3 suppressed spread of primary mammary tumors in mice to the lungs but did not suppress their growth. However, a combination immunotherapy with GARP antibody treatment could enhance the efficacy of standard chemotherapy in this situation in humans.
Unfortunately, Li also has to consider one problem with this approach: targeting GARP might also block the helpful activity of Tregs, which are designed to suppress the immune system in order to prevent autoimmune disease.
"Clinically some of the proven immunotherapies do induce some degree of autoimmunity," Li said. "When cancer is cured and patients stop immunotherapy, the autoimmune manifestations completely disappear as well." Li’s study was recently published in the journal Cancer Research.
"This discovery is fundamentally important to how TGF-beta utilizes GARP to promote cancer and down-regulate the immune system, but it also creates an opportunity for both diagnostics and therapeutics," Li said. His study provided the first evidence for GARP as a diagnostic marker for cancer in addition to an opportunity for new cancer treatments.
For more on TGF-beta signaling:
Sources: Cancer Research, R&D Systems, Medical University of South Carolina