PCR Innovation Could Improve, Mobilize Technique
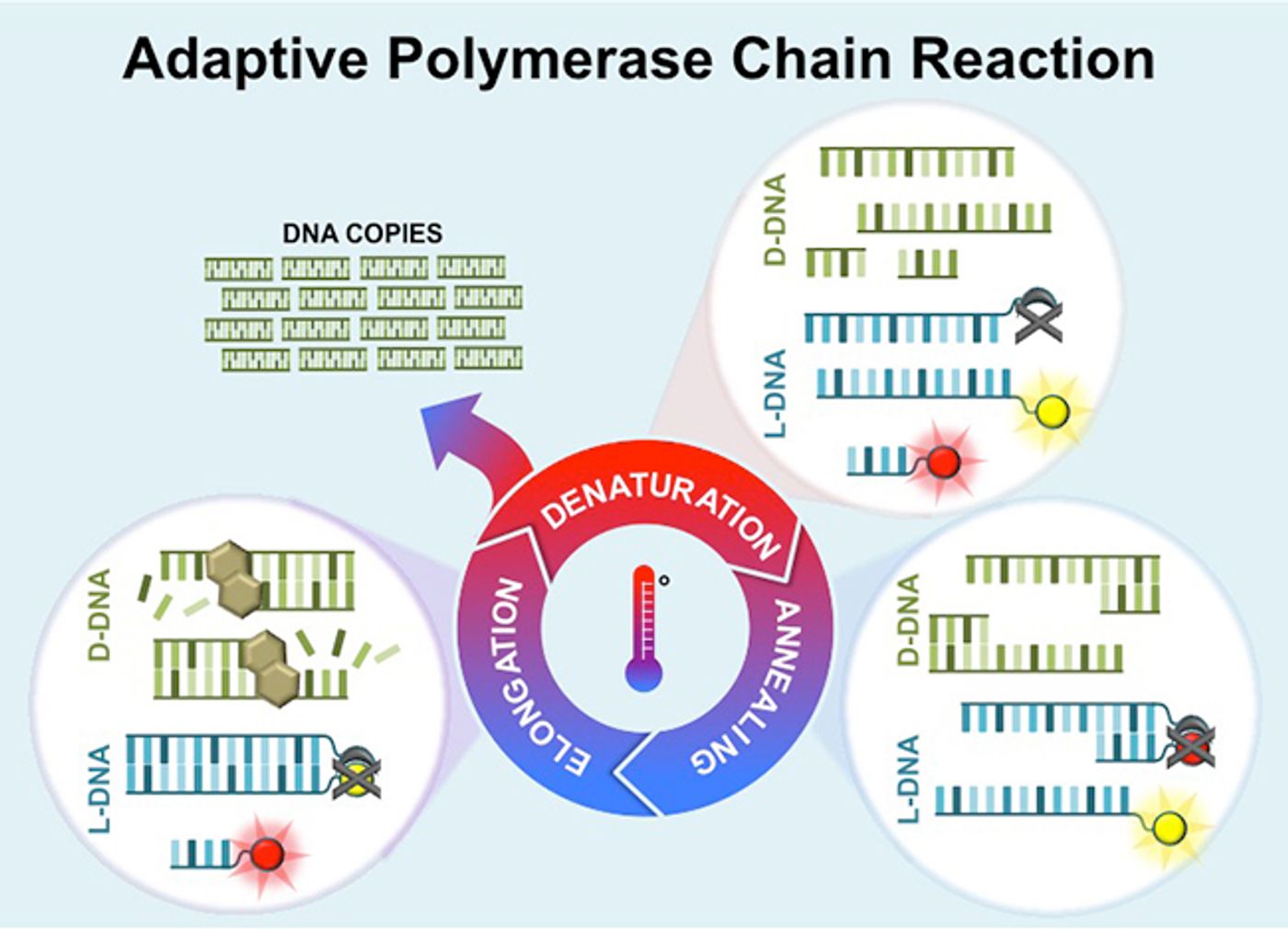
Polymerase Chain Reaction or PCR is common in research and diagnostic laboratories, performed on specialized PCR machines. But researchers at Vanderbilt University have used a method called adaptive PCR, utilizing left-handed DNA, L-DNA, to get around typical variables in PCR conditions. That could free the technique from the laboratory, as it would no longer have so many special requirements. The innovation is described in the following video and published in the journal Analytical Chemistry.
PCR is a sort of photocopier for PCR in which small pieces of DNA are used to copy DNA from a template. In typical PCR, temperature is critical. Chemicals are another important reaction variable. PCR machines need good insulation; they have to be able to cycle through precise temperatures during the reaction. Small variations in those conditions can cause the reaction to fail.
In adaptive PCR, the L-DNA is used as a monitor of what is happening at the molecular level. L-DNA is a mirror image of normal, right-handed DNA, but it does not interact in biological reactions like right-handed DNA. As such, L-DNA with a fluorescent tag can be added to a PCR sample and used as a reporter that can provide feedback about the reactions taking place.
To perform the process, a researcher would have to first get L-DNA with the same sequence as the DNA that is to be amplified. The L-DNA would be commercially available, and come with a fluorescent dye on strand, and a molecule to quench the fluorescence on its opposite strand. During a PCR reaction, double-stranded DNA molecules are separated by a high temperature during a denaturation step. In adaptive PCR the L-DNA would also separate during the denaturation step, thus separating the dye and quencher, causing an increase in fluorescence.
As PCR continues, DNA primers that are specific to the region of DNA that is to be amplified bind to the template. In adaptive PCR, left-handed primers would be used, and they also would have a dye quencher attached. Those quenchers would reduce fluorescence emanating from the L-DNA template. The reduction of signal indicates when primers have attached to most of the DNA template strands.
PCR would then continue and amplify the DNA, but since L-DNA does not participate in that step, its level remains constant the entire time.
The researchers believe that this technique has the capability to amplify DNA when normal conditions fail, and will improve PCR. They commented in a press release: “These advantages have the potential to make PCR-based diagnostics more accessible outside of well-controlled laboratories, such as point-of-care and field settings that lack the resources to accurately control the reaction temperature or perform high quality sample preparation.”
Sources: Vanderbilt University, Analytical Chemistry