Glioblastoma Treatment Combines Vaccine and Chemo Technology
A rare and lethal brain tumor more often than not leads to death not much longer than one year after diagnosis, even with treatment. However, a new combination treatment for this rare cancer, called glioblastoma, showed surprisingly promising results in a new, small study.
The new study from the Duke Cancer Institute tested the treatment in just 11 patients, so researchers are cautiously drawing conclusions. However, the combination therapy, which included an experimental vaccine alongside a “dose-intensified” version of an approved chemotherapy, at first glance seems safe and effective.
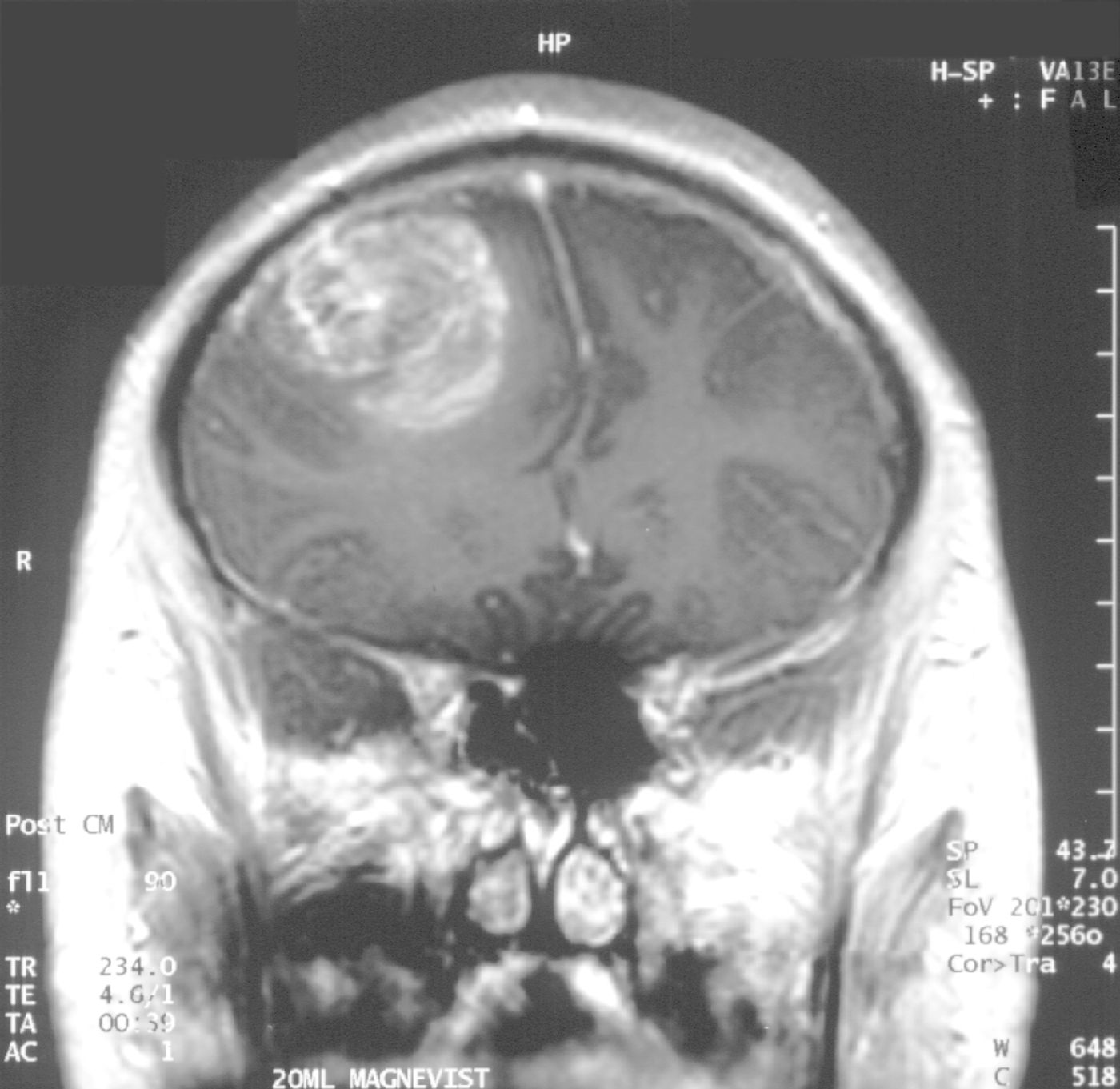
Glioblastoma patients make up about 15 percent of all those suffering from primary brain tumors. These highly malignant masses are usually discovered in the brain’s cerebral hemispheres, and although this cancer rarely metastasizes, the tumor cells grow rapidly and aggressively. Glioblastoma increases brain pressure, which can cause headaches, nausea, vomiting, and drowsiness. The cause of glioblastoma is unknown.
Introduced in the Duke study is an investigational immunotherapy approach consisting of a dendritic cell vaccine that very specifically targets cytomegalovirus proteins. These proteins are abundant in and unique to glioblastoma tumors, so such a treatment leaves surrounding healthy brain cells untouched.
The dendritic cell vaccine is paired with an approved chemotherapy drug called temozolomide, which is applied in a higher dose. According to the researchers, using a dose-intensified version of the drug boosts the immune system and enhances the efficacy of the dendritic cell vaccine paired with it.
"Our strategy was to capitalize on the immune deficiency caused by the temozolomide regimen," explained lead author Kristen Batich, MD, PhD. "It seems counter-intuitive, but when the patient's lymphocytes are depleted, it's actually an optimal time to introduce the vaccine therapy. It basically gives the immune system marching orders to mount resources to attack the tumor."
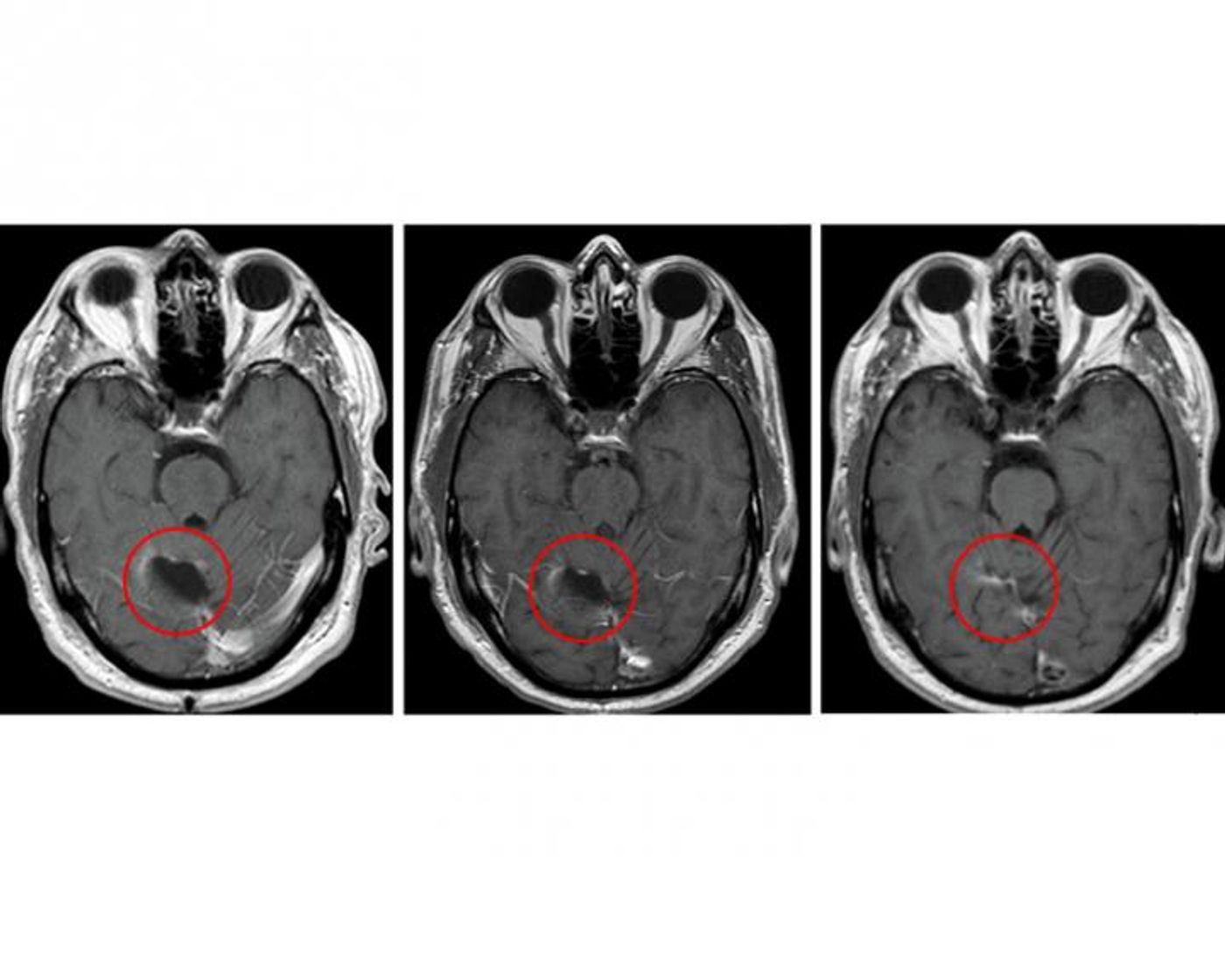
Researchers closely monitored the 11 glioblastoma patients who received the new treatment, and the only side effects seen were expected in connection to temozolomide. Four out of the 11 study participants survived for more than five years following treatment.
"While not a controlled efficacy study, the survival results were surprising, and they suggest the possibility that combining the vaccine with a more intense regimen of this chemotherapy promotes a strong cooperative benefit,” Batich explained.
With promising results already under her belt, Batich plans to conduct larger, controlled clinical trials with this new investigational drug, hoping for even more favorable outcomes. The present study was published in the journal Clinical Cancer Research.
Sources: American Brain Tumor Association, Duke University Medical Center