Treating A Rare Immunological Disease with Mepolizumab
There's a new standard treatment for a rare immunological disease: mepolizumab. No need for an approval process, though - the drug is already approved by the U.S. Food and Drug Administration (FDA) to treat another disease.
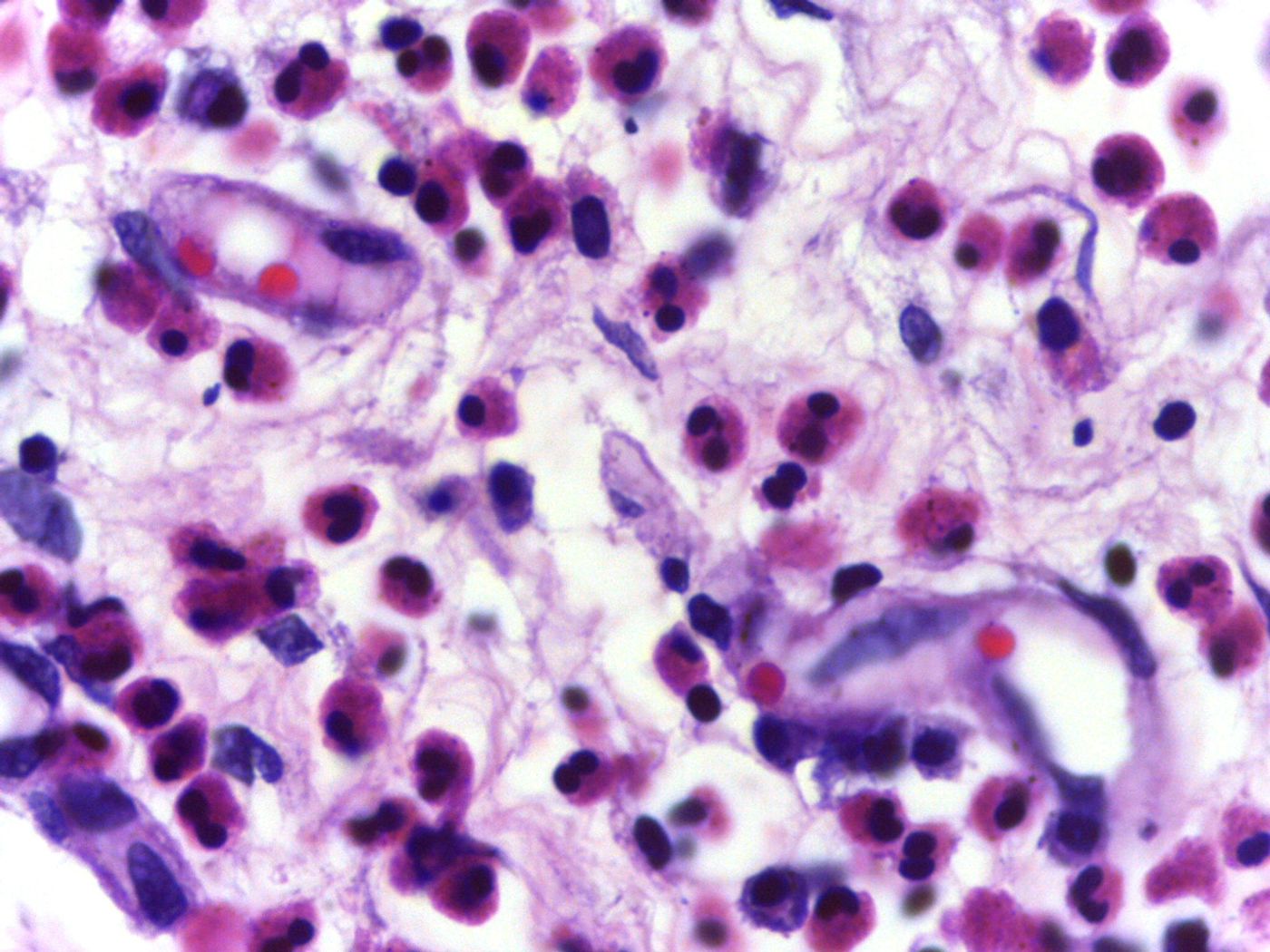
The identification of a new treatment is promising news for people with the rare disease, called eosinophilic granulomatosis with polyangiitis (EGPA). Previously known as Churg-Strauss Syndrome, EGPA is characterized by excessive proliferation of eosinophils, a type of proinflammatory white blood cell involved in multiple immune processes, including allergic reactions. As the eosinophil population grows, cells start to collect in the blood vessel walls, meanwhile releasing toxic, inflammatory proteins that damage tissues around them.
An individual developing EGPA will first experience only severe asthma, but as time goes by, more blood flow is restricted, and more tissues are damaged, multi-organ damage will occur. Prior to mepolizumab, the only standard treatments for EGPA were oral steroids, taken regularly, and occasionally immunosuppressants. However, both options are accompanied by adverse side effects, relapses are common, and treatment doesn’t always work for some EGPA patients.
Mepolizumab was originally approved by the FDA to treat serious cases of eosinophilic asthma; the drug is a humanized monoclonal antibody of immunoglobulin G, and it targets an immune chemical called interleukin-5 to treat asthma.
The new phase 3 Clinical Trial, from the National Institute of Allergy and Infectious Diseases, examined the improvements made in patients when mepolizumab is added to steroid treatments for EGPA. Researchers conducted a double-blind experiment of 136 EGPA participants, where half of the study’s participants received mepolizumab and half received a placebo. All of the study’s participants either did not respond at all to steroids or relapsed after taking oral steroids.
Regardless of whether or not they also took immunosuppressants, nearly one-third of people receiving mepolizumab remained in remission for at least 24 weeks, but only three percent of the placebo participants remained in remission for that long.
The next step for getting mepolizumab into the hands of people struggling with EGPA: What biological markers can distinguish between people who would benefit from mepolizumab and those who would not?
The present study was published in the New England Journal of Medicine.
Sources: NIH/National Institute of Allergy and Infectious Diseases, Expert Review of Clinical Immunology, Cincinnati Center for Eosinophilic Disorders