Yellow Fever Virus is No Match for This Immune Signaling Pathway
A vaccine for yellow fever was first developed in 1937, using a strain of the yellow fever virus that was just a few amino acids away from being a carbon copy of the actual infectious virus. The vaccine all but eradicated yellow fever, with isolated jungle epidemics still occurring in some parts of the world. Now scientists from Princeton University are using the vaccine to study the intricate details of the antiviral immune response.
It runs in the family
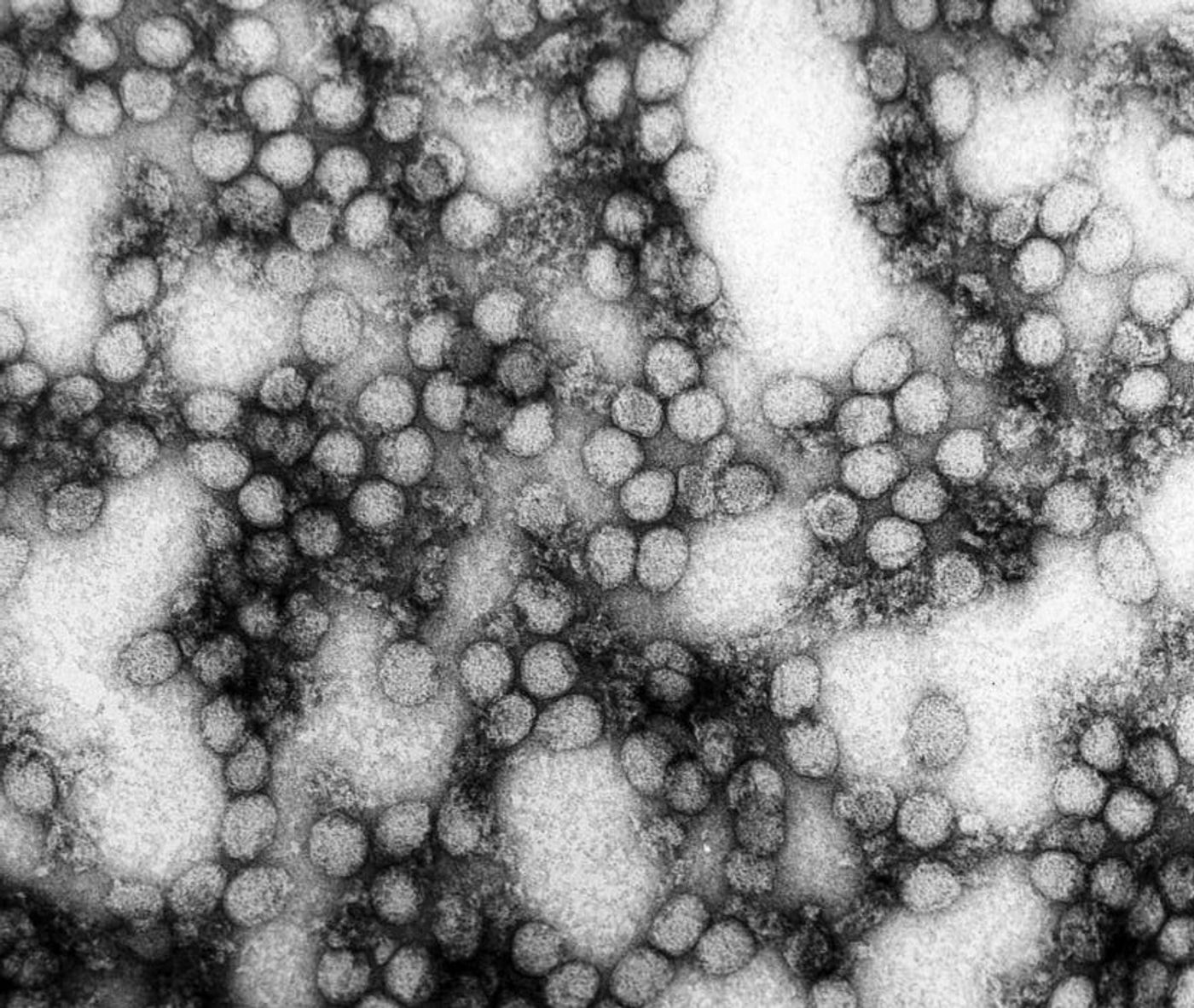
The yellow fever virus, a flavivirus, comes from the same family of viruses as West Nile, Dengue, and Zika. By studying the immune signaling pathway that provides the foundation of an immune response against yellow fever, scientists could potentially unveil new ways to fight these other flavivirus family members.
The vaccine against yellow fever, called YFV-17D, is live-attenuated, which means that the virus is still viable and triggers an immune reaction, but scientists have taken away or reduced the parts that make the virus dangerous. Since its initial development early in the 20th century, YFV-17D has been proven to successfully stir up the immune system against yellow fever viral proteins in case a person actually came into contact with yellow fever.
Princeton University scientists began by using mouse models, infected with YFV-17D, to associate immune cytokines called interferons (IFN) with the antiviral immune response. There are three types of IFN possessed by both mice and humans, and each type has a specific receptor that it needs to bind to be active. All IFN receptors are common in the body.
-
Type I, IFN-α/β receptor
-
Type II, IFN-γ receptor
-
Type III, IFN-λ receptor
Interestingly, mice without IFN-α/β receptors die if infected with actual yellow fever, but not if just given the vaccine. Either way, Type II IFN signaling makes up for lacking Type I IFN in both situations. The only component missing from the equation was the role played by Type III IFN signaling.
What happens when mice don’t have IFN-λ, the receptor for Type III IFN signaling?
Mice lacking Type III IFN signaling were given the vaccine, and it didn’t seem that Type III IFN was needed for an adequate immune response because the mice dealt with the virus without a problem. However, those missing both Type I and Type III signaling did not fare as well, indicating Type III signaling is needed after all, if just in an assistive role.
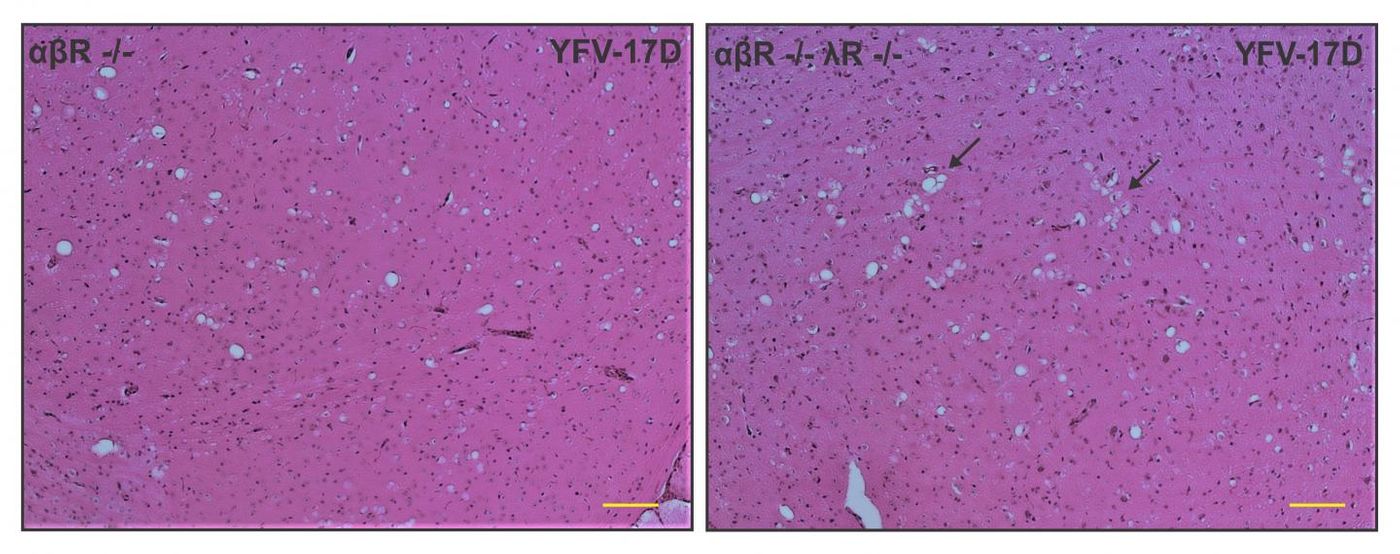
Princeton University researchers looked closer, examining viral levels in different types of tissue after vaccination. They saw low viral loads in wild-type (normal, healthy) mice and and those lacking Type III IFN receptors. But in mice lacking both Type I and Type III signaling, high viral loads that continued to increase.
Left panel: Loss of Type I IFN signaling leads to active replication of the attenuated YFV strain (YFV-17D), which is accompanied by viral invasion of the brain and damage to brain tissues (spongiosis). Right panel: The additional loss of Type III IFN signaling in Type I IFN-deficient mice impairs the integrity of the blood-brain-barrier and alters immune cell function, which aggravates spongiosis and is ultimately lethal. Credit: Florian Douam and Alexander Ploss
The damage done when mice lacked Type III IFN signaling seemed to come from increased viral loads in the brain, leading to spongiosis (swelling) or encephalitis (inflammation). But why?
According to past research, Type III IFN signaling involves epithelial cells in the blood-brain barrier (BBB), which serves to protect the central nervous system from pathogens like yellow fever. Without Type III IFN signaling, the integrity of the BBB faltered just enough to let the virus in. Researchers discovered this connection in a model of West Nile Virus, which comes from the same family as the yellow fever virus.
With this finding in mind, researchers from the present study confirmed that the BBB was similarly weakened in mice lacking Type I and Type III IFN signaling after yellow fever vaccination. Additionally, they found that Type III IFN signaling is vital for the activation of T cells after vaccination, immune cells at the center of the adapted immune response.
Now that they know monumentally more about the role of IFN in the antiviral immune response to yellow fever, the research team plans on moving their work to the context of primates, bringing them closer to applying their knowledge in the clinic to bolster the arsenal of tools available to fight all types of flaviviruses.
The present study is published in the journal mBio.
Sources: Princeton University, The Journal of Experimental Medicine, Basic Neurochemistry: Molecular, Cellular and Medical Aspects