As If Superbugs Weren't Bad Enough, Here's Another Reason Not to Overuse Antibiotics
Ever since penicillin was discovered in 1928, bacteria have been fighting back against every single antibiotic we develop. Unfortunately, we’re helping bacteria become resistant by being careless with the use of antibiotic drugs, and a recent study from the University of Virginia shows that in addition to creating superbugs, the overuse of antibiotics is hurting the immune system’s ability to fight pathogens.
Contributions to Antibiotic Resistance
When you’re given a prescription for antibiotics to treat a bacterial infection, your doctor always stresses the importance of taking all of the pills, even if you start to feel better. If you don’t kill all of the bacteria, the infection could return, and only the strongest and most resilient bacteria would be the ones surviving and multiplying. For the same reason, it’s important for doctors not to over-prescribe antibiotics and to limit the use of antibiotics in commercial agriculture.
Now, there’s another reason to use antibiotics carefully and appropriately.
While studying amebic colitis and gut microbiome, the population of healthy bacteria living in parts of the body like the gastrointestinal (GI) tract, researchers found that a reduction in the diversity of gut microbes due to excessive or inappropriate antibiotic use impairs the ability of immune cells called neutrophils to respond to pathogenic threats in the body.
Neutrophils: vital first-responders
Neutrophils lead the attack when pathogens enter the body. They absorb and digest pathogens, a process called phagocytosis. "We found that antibiotic disruption of the natural microbes in the gut prevented this from happening properly, leaving the gut susceptible to severe infection,” explained Virginia’s Koji Watanabe, PhD.
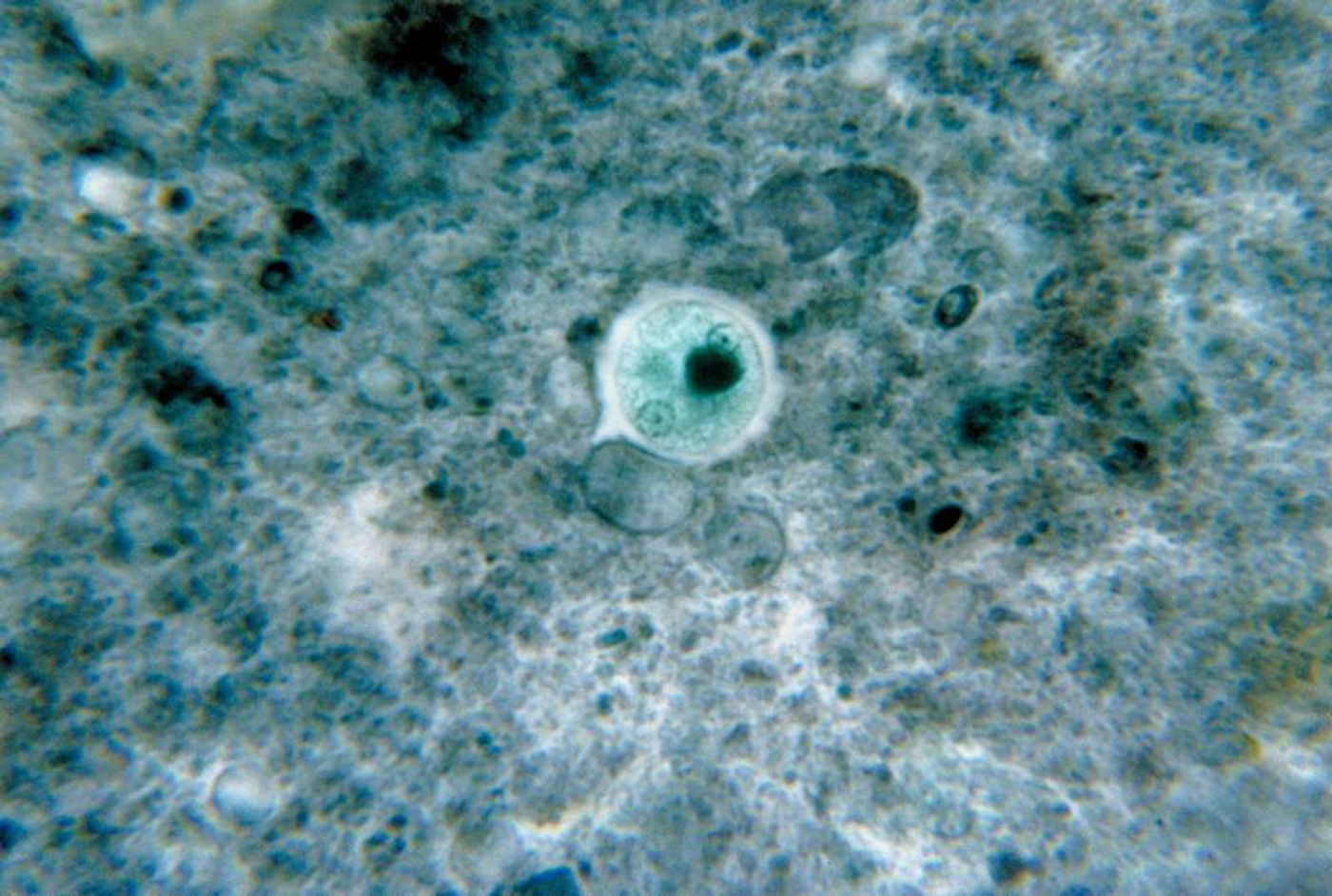
Watanabe and the other researchers were studying amebic colitis, a disorder of the GI tract most common in underdeveloped countries. The disease is caused by a protozoan parasite called Entamoeba histolytica. There are approximately 40 million cases of amebic colitis reported worldwide every year. The parasite attaches to the lining of the digestive tract, poking holes in the tissue to travel to and infect other parts of the body.
During a study in Bangladesh slums, researchers collected stool samples from children. Those with the most severe cases of the amebic colitis were also the ones with the least diverse gut microbiomes. Later, in mice models, researchers saw that antibiotic-directed disturbances in the gut microbiome reduced neutrophil activity to the point where neutrophils wouldn’t respond to the immune messenger chemicals signaling an infection. Additionally, the intestinal barrier that separates the GI tract from the rest of the body was “compromised.”
This is another important reason not to use antibiotics unless they are clearly needed," explained Bill Petri, MD, PhD, chief of the Division of Infectious Diseases at the University of Virginia. "Unwise use of antibiotics not only increases the risk of multi-drug resistant bacteria and the risk of C. difficile infection but also impairs white blood cell function."
The present study was published in the journal PLOS Pathogens.
Sources: University of Virginia Health System, PubMed Health, The Internet Journal of Advanced Nursing Practice, National Academies Press