Neuro-Immune Dialogue Burns Belly Fat
A unique interaction between immune cells and the nervous system could explain why older people have more belly fat - and why they have a hard time getting rid of it. From Yale University and other collaborators, scientists discovered a new subtype of immune cell and a connection to a specific neurotransmitter that explains how the message - burn fat - gets lost in translation.
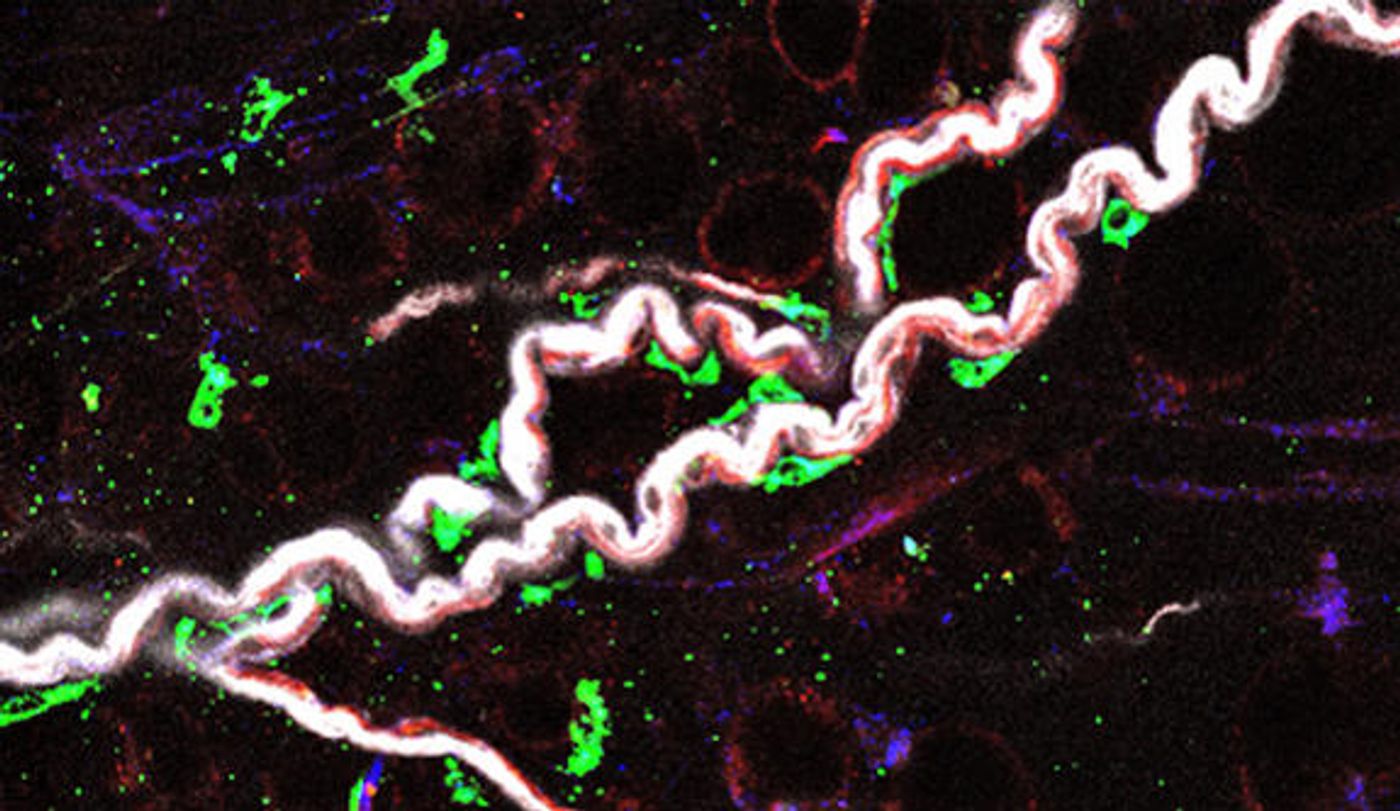
Credit: Ziba Kashef, Yale University
The older you get, the more belly fat you tend to collect, an affliction that occurs independently of your actual body weight. To make matters worse, older people are less effective at burning the growing amount of belly fat than younger people. Researchers from Yale University want to know why.
First, Yale researchers identified a new subtype of macrophages that become increasingly inflammatory over time. They live in the nerves of belly fat, preventing important chemical messengers of the nervous system, neurotransmitters, from communicating effectively. Researchers isolated and analyzed these macrophages, taken from the fat tissue of both young and old mice.
"We discovered that the aged macrophages can break down the neurotransmitters called catecholamines, and thus do not allow fat cells to supply the fuel when demand arises,” explained study leader Vishwa Deep Dixit.
Neurotransmitters transfer signals between neurons. Dopamine and norepinephrine are both catecholamines, neurotransmitters employed by the nervous system that play a primary role in motor control, cognition, emotion, memory processing, and endocrine modulation.
Next, the researchers discovered that lowering the activity of the NLRP3 inflammasome, which mediates the inflammatory response, brought catecholamines back up to speed, directing fat breakdown in aged macrophages as if they were macrophages in young mice. “Immune cells talk to the nervous system to control metabolism,” Dixit explained.
Their last experiment involved anti-monoamine oxidase-A (MAOA) drugs, which currently exist on the market to treat depression. The MAOA enzyme is present in higher levels in aged macrophages than in younger macrophages, and blocking MAOA revitalized the process of fat metabolism.
Going forward, Dixit and the researchers from this study will continue to study the intricacies of the “neuro-immune dialogue” in influencing fat metabolism and inflammation. As far as anti-MAOA drugs to target unresponsive belly fat, they will need to test its efficacy in humans as well as the safety of its use.
In addition, Dixit wonders: What other benefits could there be to controlling inflammation specifically in aging immune cells?
The present study was published in the journal Nature.
Sources: The Journal of Investigative Dermatology, Yale University