How the Gut Microbiome Controls the Intestinal Immune System
The microorganisms that live in our gastrointestinal system (known collectively as the gut microbiome) have many important functions, including helping us digest food and get the right nutrients. Researchers at Brown University have now used a mouse model to show that the gut microbiome exerts control over the immune system of its host, so the immune system will not attack the bacteria in the microbiome. They determined that gut bacteria can regulate levels of Vitamin A, which protects the microbes from an immune response. The findings, which have been reported in Immunity, may help us understand autoimmune diseases and how to treat them.
"A lot of these diseases are attributed to increased immune response or immune activation, but we've found a new way that bacteria in our gut can dampen the immune response," said Shipra Vaishnava, an assistant professor of molecular microbiology and immunology at Brown. "This research could be critical in determining therapies in the case of autoimmune diseases such as Crohn's disease or other inflammatory bowel diseases, as well as vitamin A deficiency."
Most of the roughly 100 trillion bacteria in the gut microbiome are beneficial to the host, which have evolved to live in a community within the gut. These microbes coexist peacefully and can coordinate against intruders. In humans and mice alike, the bacteria phyla Firmicutes and Bacteroidetes are the primary players in the gut microbiome.
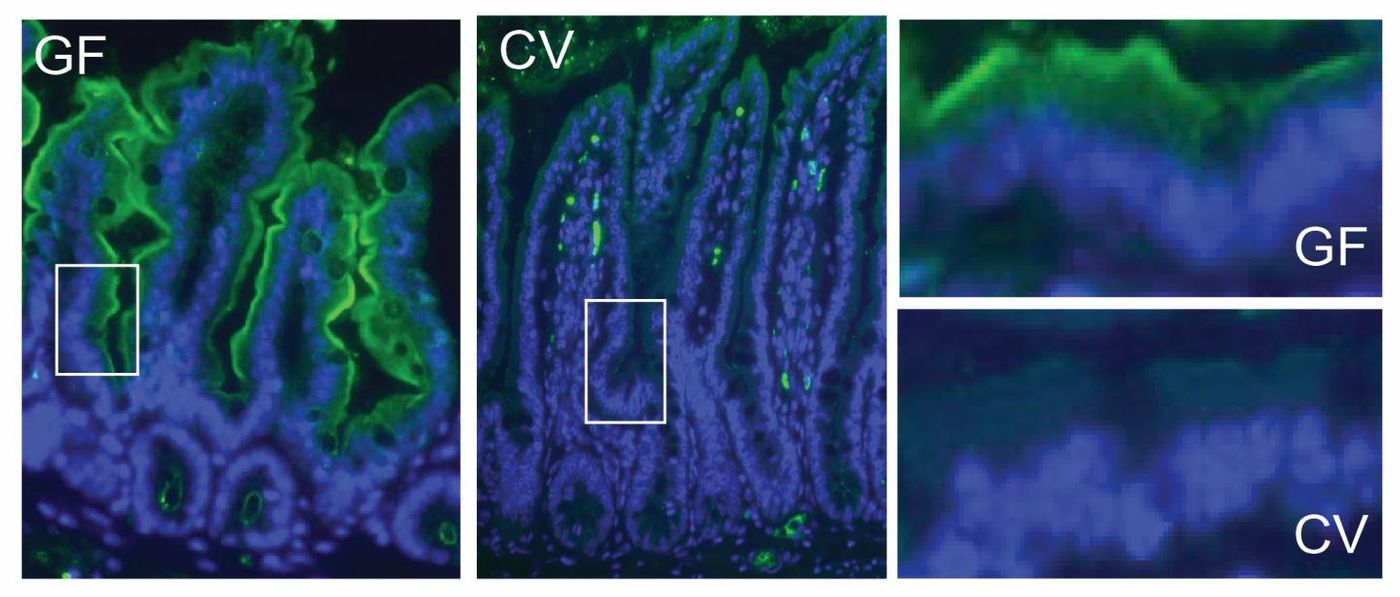
In this work, the researchers found that Firmicutes bacteria, especially those classified as Clostridia, can lower the expression level of a protein called Rdh7, retinol dehydrogenase. Rdh7 can change dietary vitamin A into its active form, called retinoic acid, noted Vaishnava. Clostridia can also encourage the storage of vitamin A in the liver. The researchers expect that although these findings were in mice, they will apply to humans too.
When mice were engineered to lack Rdh7 in their intestines, they had less retinoid acid than normal mice, as expected. The modified mice also had fewer immune cells that produce a signal called IL-22, which helps orchestrate the antibacterial response to gut microbes. Other parts of the immune system were unaltered in the edited mice, however. That suggests that Rdh7 only functions in controlling the antimicrobial response, said Vaishnava. Now, the researchers are working to find out how Clostridia suppresses Rdh7, and why it’s so important.
Understanding the relationship between the gut microbiome and the regulation of the immune system response might help us treat diseases like Crohn’s, said the researchers. Clinical studies show that disruptions in the host-microbiome relationship causes inflammation, noted Vaishnava.
"The role of vitamin A in inflammation is context-dependent and is very hard to tease apart," Vaishnava said. "A change in vitamin A status and vitamin A metabolic genes coincides with inflammatory bowel diseases, but we don't know if this promotes inflammation or not. We hope that adding our finding -- that bacteria can regulate how vitamin A is being metabolized in the intestine or stored -- could help clarify why the field is seeing what it is seeing."
This work may also help address vitamin A deficiency, a problem for children in Africa and Southeast Asia. They may benefit from bacterial treatments since microbes play a role in the absorption and storage of vitamin A.
"Both our diet and the bacteria in our gut are critically linked in regulating how our immune cells behave," Vaishnava said. "Finding what those links are at a molecular level is important to figuring out how we could use either diet or bacteria, or both of them together, to have a therapeutic effect in inflammatory or infectious diseases."
Check out two lectures about the gut microbiome and host immunity. The one above is by Johns Hopkins Medicine, and the one below is from the NIH.
Sources: AAAS/Eurekalert! Via Brown University, Immunity