Boosting the Efficacy of Vaccines Against Type B Flu
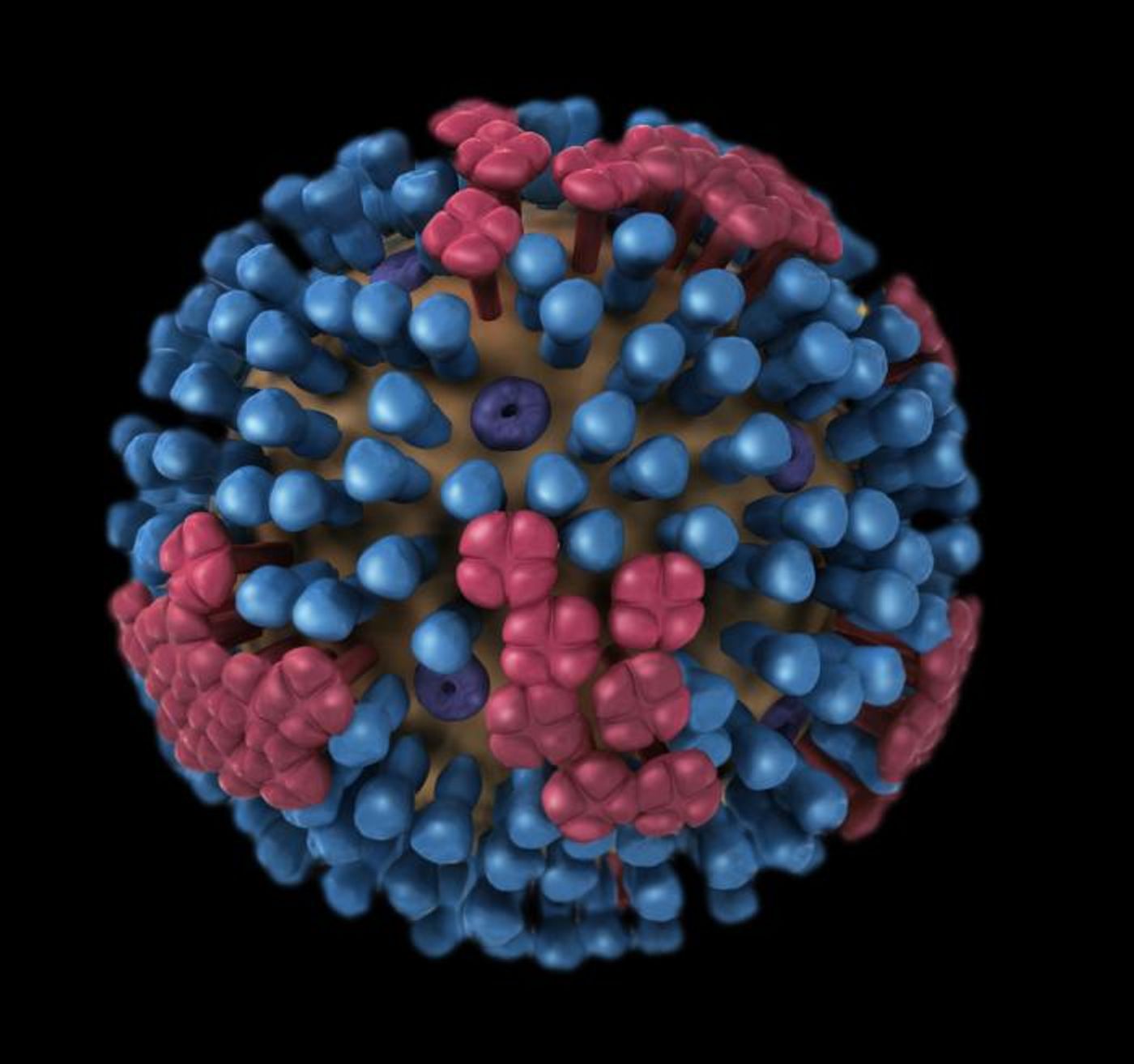
There are four different classification of influenza virus - type A, B, C, and D. The A and B types usually cause seasonal flu epidemics in the United States every winter. When new types of influenza A emerge, they have the potential to trigger an epidemic, and can be subcategorized by strain. Type B viruses aren’t classified into subtypes; they are simply classified by strain and lineage. Although type B influenza is not as common as type A, it still threatens the health of the young and old, and was to blame for most flu deaths among children during the 2012 - 2013 season according to the Centers for Disease Control.
Researchers have now confirmed that there may be a straightforward way to improve the efficacy of vaccines in the fight against type B flu. The research has been reported in mBio. A molecule called neuraminidase (NA), which is present in current vaccines, stimulates the immune system to generate antibodies that could protect against an array of influenza type B viruses. Previous research has linked flu protection and NA antibodies; they may halt the spread of infection.
"Targeting this type of vaccine response can help us develop a universal vaccine," said influenza virologist Luis Martinez-Sobrido, Ph.D. He and immunologist James Kobie, Ph.D., co-led the study at the University of Rochester.
The surface of the flu virus carries antigens - they trigger the immune system to produce antibodies that will fight the virus, and are used in vaccines. Most of these antibodies target common flu antigens called hemagglutinins (HA). They can change or mutate from year-to-year in flu viruses; that’s why new flu vaccines must be made annually (as described in this video).
NA antigens are also found on the surface of the flu virus. While NA and HA antigens are both on the viral surface, there are about four times as many HA proteins. The NA proteins mutate at a much lower rate though, and a vaccine that targets them may be effective for several years since they don't change much, noted Martinez-Sobrido.
In this work, the scientists used blood samples from participants that had gotten a quadrivalent flu vaccine for the season. NA antibodies against type B flu particles were found to be produced after the vaccine was used. Additional work identified six antibodies that target type B flu NA particles.
"These are broad antiviral agents that work against a wide range of influenza B strains," said Dr. Kobie.
The researchers showed that the vaccine could stop viral particle release in human cells, and prevent and treat infections in a mouse model.
They were also able to obtain a bone marrow sample from a study participant one year after they’d received the quadrivalent vaccine. They found that antibodies that had anti-viral activity were still being produced.
Current vaccines may not provide enough stimulation to trigger sufficient productions of antibodies against NA, but this work shows that it may be possible to do so in the future.
"Seasonal vaccines are good in protecting us against influenza," said Martinez-Sobrido, "but there is room for improvement. By tuning a vaccine to elicit more of an NA-related response like the one found in our studies, we could go further in developing a universal vaccine."
Sources: AAAS/Eurekaert! via American Society for Microbiology, mBio