Neurons in the Gut Can Detect Salmonella & Protect Against Infection
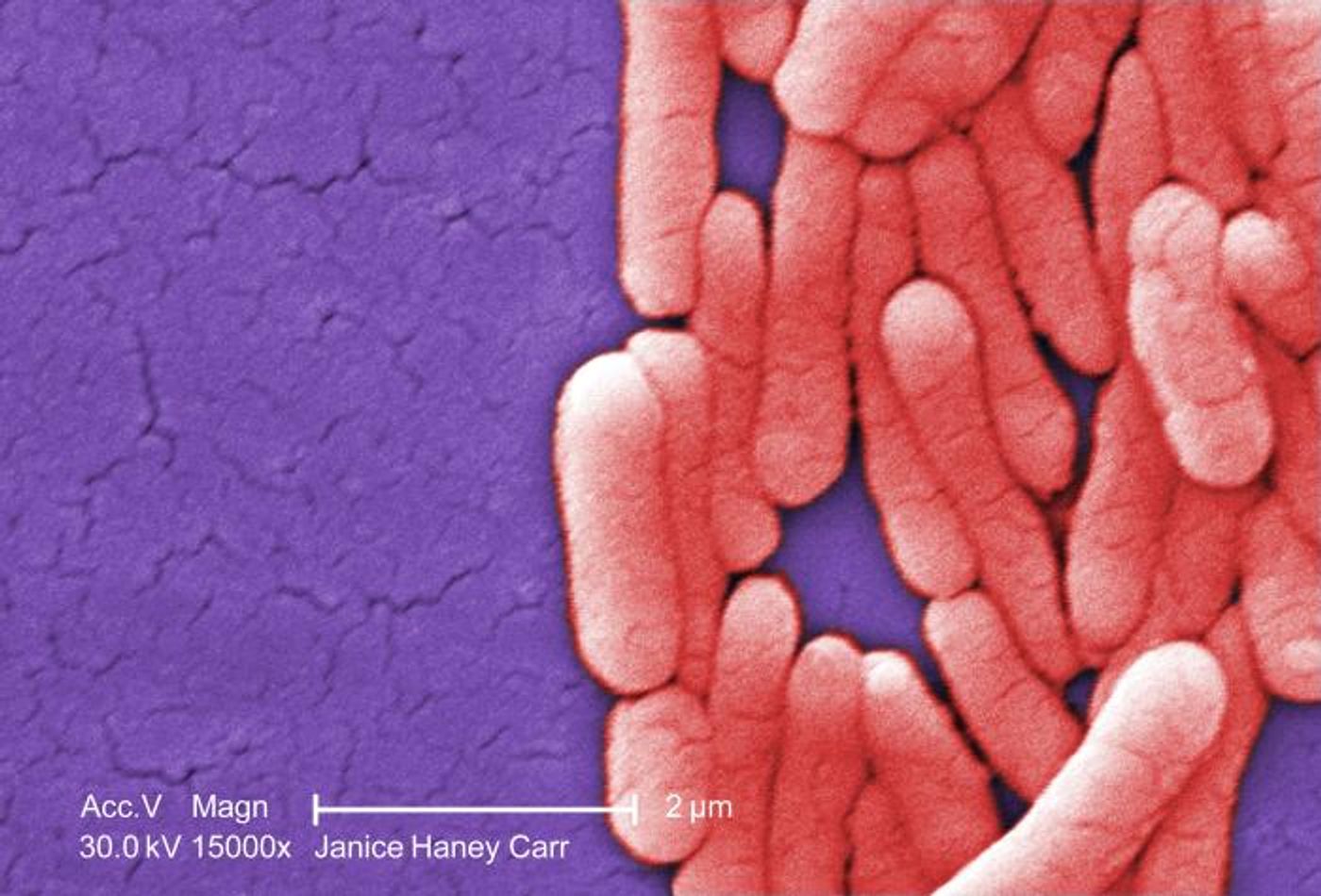
Nerve cells act as critical sensors for the human body, detecting all kinds of stimuli and coordinating responses. Now researchers have identified a protective role for neurons; in the guts of mice, neuronal cells were found to be actively protecting against Salmonella infection. Salmonella microbes are responsible for around 25 percent of bacterial diarrhea globally. Gut neurons that sense pain and reside in the small intestine were found to be activated by Salmonella bacteria. The nerve cells are then able to prevent infection, stopping this microbe from spreading through the body. The findings have been reported in Cell.
"Our results show the nervous system is not just a simple sensor-and-alert system," said the lead investigator, neuro-immunologist Isaac Chiu, an assistant professor of immunology in the Blavatnik Institute at Harvard Medical School. "We have found that nerve cells in the gut go above and beyond. They regulate gut immunity, maintain gut homeostasis, and provide active protection against infection."
The neurons that are triggered by Salmonella sit beneath masses called Peyer's patches, which contain M cells that open and close, regulating the flow of stuff in and out of the small intestine. Dangerous bacteria, including Salmonella, use M cells to get into the small intestine.
In this work, the researchers compared normal mice to mice that had been genetically engineered to lack gut neurons and mice in which gut neurons were chemically disabled. They found that when Salmonella is in the small intestine, normal gut neurons release a molecule called CGRP to reduce the number of M cells so there aren't as many entry points for the microbe. The mechanism is unclear, but CGRP also seems to boost the number of protective microbes. In animals without normal gut neurons, Peyer's patches were found to be loaded with Salmonella bacteria, and didn't have as many protective microbes. These mice had higher rates of Salmonella sickness, which was also more widespread compared to normal mice.
"It is becoming increasingly clear that the nervous system interacts directly with infectious organisms in various ways to affect immunity," Chiu said. "Bacteria literally do get on our nerves."
The gut has been called the second brain because it contains so many neurons, called the enteric nervous system. Previous work by the Chiu lab has shown that infections impact both the immune and nervous system; lung neurons can affect the immune response to lung infection, and microbes that cause flesh-eating disease can exploit nerves to reduce the immune response to infection.
"Our findings illustrate an important cross-talk between the nervous system and the immune system," said the first author of the study Nicole Lai, a research fellow in immunology in the Chiu lab. "It is clearly a bidirectional highway with both systems sending messages and influencing each other to regulate protective responses during infection."
"If you think about it, the nervous system's involvement in immunity is an evolutionarily smart way to protect the gut from infection by repurposing an existing feature," Chiu said.
This work, said the scientists, can also explain why opioid use is linked to higher rates of infection. "If you dial down nerve signaling in an effort to reduce pain, you may be inadvertently also dampening their protective abilities," Chiu said. "Our observations support that idea."
The study may also open up new therapeutic avenues; altering nerve signaling might increase immunity or reduce inflammation in the gut.
"The idea would be that if we could somehow stimulate these protective gut neurons or mimic their activity with a drug, we could activate the immune response and increase the body's ability to fend off infection," Chiu said.
Sources: AAAS/Eurekalert! Via Harvard Medical School, Cell