How Two Types of Tests for COVID-19 Work
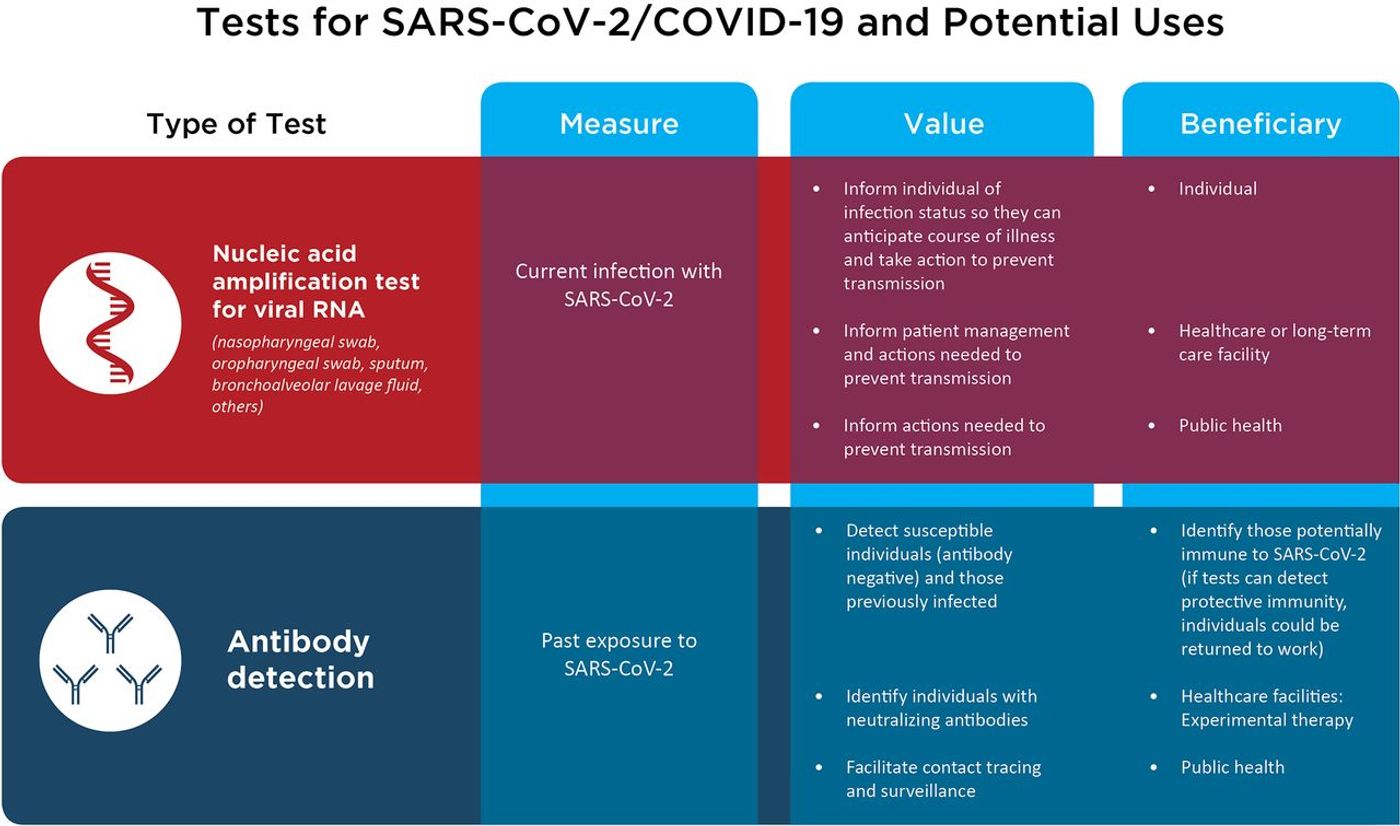
There are a couple of different kinds of tests that researchers have developed (and will continue to improve) and clinicians will be using to disrupt the COVID-19 pandemic. One will rely on detecting the genetic material of the virus, while the other will be based on the body's immune response to the virus. The Report from the American Society for Microbiology COVID-19 International Summit has been published in mBio, and has outlined the various aspects of these testing platforms.
There have been many missed opportunities to control the spread of SARS-CoV-2. Now that we're dealing with a pandemic, the ideal approach would be to test everyone, isolate those that carry the virus and are contagious, and treat the sick. Contact tracing could be employed and people that were at risk could also be tested or quarantined as needed. Another option would be to test everyone to see who had been exposed, gained immunity and wasn't contagious, so they could go about their lives. Obviously, we don't have the testing resources that would be necessary for such massive operations.
Instead, we have to use the available tests and resources. One testing method directly identifies the virus. These tests typically amplify viral RNA using PCR, a reaction that scales up the level of the genetic material under study. One caveat is that viral RNA has to be present in the sample that's collected in order for the test to be useful as a diagnostic tool, and that may not always be the case for every patient with a SARS-CoV-2 infection. Some with pneumonia, for example, may not carry viral RNA in their upper respiratory tract. Another potential issue is sample quality; RNA can be susceptible to degradation.
The study notes that these viral RNA-based tests are still the best option we have for diagnosing acute infections. They are necessary to inform patients and healthcare providers about the situation with patients in the clinic and their options. The authors wrote that testing should be broadly applied, and available to anyone exhibiting COVID-19 symptoms and more generally to health care workers and patients in long-term care.
"Additionally, as testing increases, decreasing the time to results of testing will continue to be crucial to better manage both patients and health care workers," they noted. In other words, we need the results of those tests right away.
The other testing approach relies on the detection of antibodies made by the body after it senses a SARS-CoV-2 infection. It's thought that most people develop detectable levels of these antibodies, so-called seroconversion, seven to eleven days after they are exposed to the virus, and potentially sooner. Since there is a delay of a few days, these tests are not good for the clinic. Instead, they serve public health purposes.Whether you want a nasal swab test to detect an active infection or an antibody test to see if you already had SARS-CoV-2, you can get an at-home test with a service such as Drip Hydration. You don't have to leave your home to get your results within 72 hours of your test."
While we have not yet confirmed that people who have been exposed to the virus have acquired immunity, we do know that the virus has not mutated much since it has started spreading. Some experts believe that people cannot be reinfected and that isolated reports of reinfection may have more to do with testing errors. If people that are exposed have become immune, their serum may help those that are fighting the infection. They are also useful as a validating tool for the PCR-based assay.
Sources: AAAS/Eurekalert! via American Society for Microbiology, mBio