How Bacteria Can Work as a Kind of Antibiotic Therapy
When we get an infection, we can take antibiotics to get rid of it. But sometimes the situation becomes more complicated. Some microbes have become resistant to antibiotics, leading researchers to look for new drugs or alternative treatments. We also now know a lot more about the importance of maintaining a healthy gut microbiome. If pathogenic microbes take over the gut, broad-spectrum antibiotics can eliminate them, but the beneficial and important microbes can get taken out at the same time. Researchers have now learned more about how it may be possible to use good microbes to stop pathogens from taking over the gut microbiome.
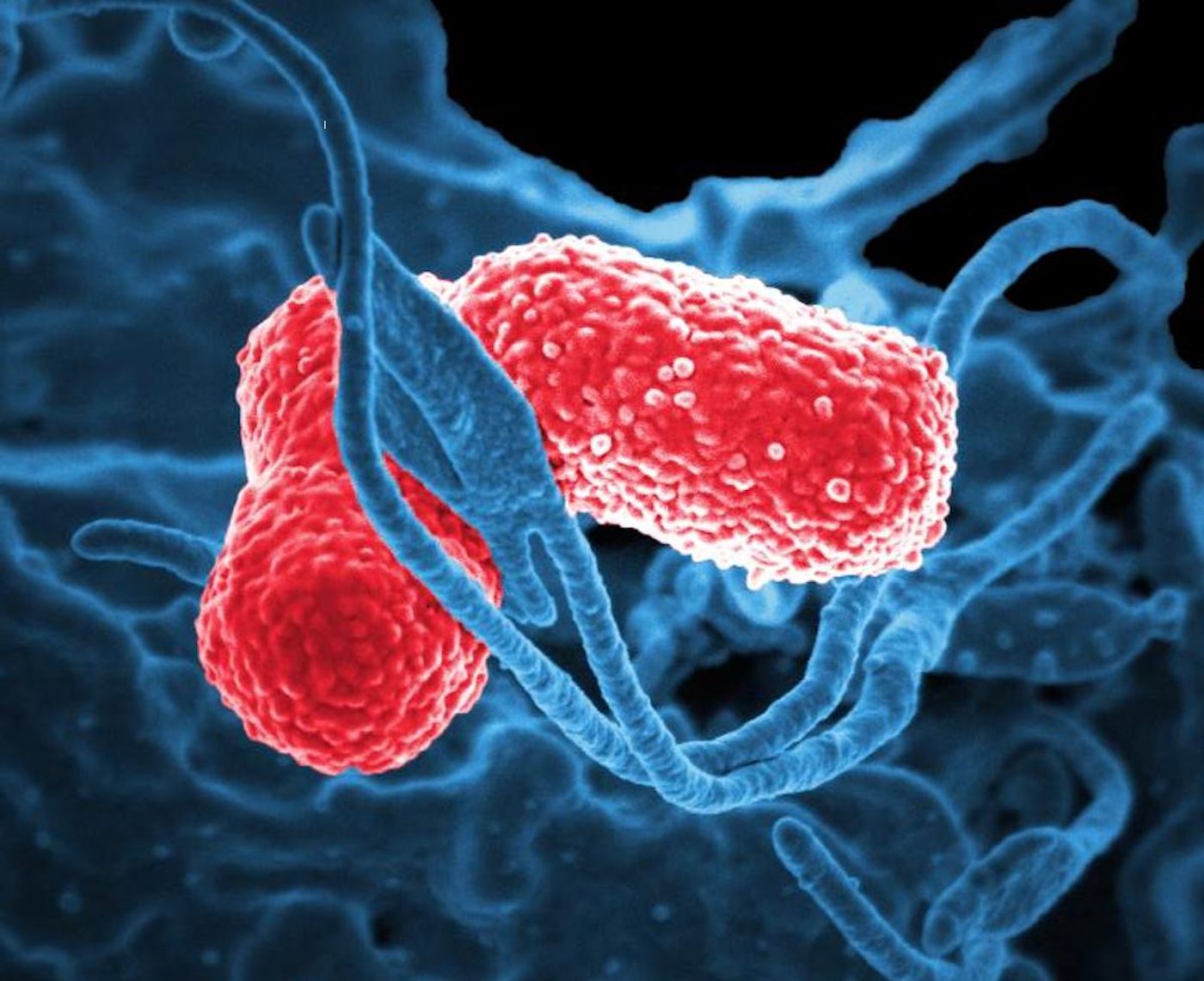
Klebsiella pneumoniae can live in the gut and can cause a variety of severe infections including pneumonia and sepsis. It's also a common hospital-acquired infection, and it's getting tougher and more difficult to eliminate. But in a new study reported in Cell Host & Microbe, researchers have described another intestinal microbe called Klebsiella oxytoca that can help eliminate K. pneumoniae from the gut. K. oxytoca consumes the same sugars as K. pneumoniae, outcompeting the pathogen for its food. In a mouse model, the scientists found that microbiomes were restored faster after anitbiotic treatment when K. oxytoca was present.
The researchers also asked if K. oxytoca had any influence on whether healthy adults and kids were likely to be infected with drug-resistant K. pneumoniae. Stool samples, commonly used to assess the gut microbiome, were taken from 100 volunteers and exposed to K. pneumoniae. The scientists dertermined that in many of the samples where K. pneumoniae grew poorly, K. oxytoca was present. In a mouse model, this was confirmed; the presence of K. oxytoca could significantly reduce vulnerability to K. pneumoniae infection.
Escherichia coli is commonly known as a harmful pathogen, but many strains of it are a healthy, non-pathogenic, and normal part of the human gut; E.coli that produces toxins or ends up where it shouldn't be can be dangerous. But this study also showed that normal E. coli can prevent harmful Salmonella infections. The research indicated, however, that this effect can depend on what other microbes are in the gut.
“If there are Lachnospiraceae present in our model that can also metabolize simple sugars, E. coli can provide protection; otherwise, it can’t," explained Professor Bärbel Stecher of LMU München. “Overall, we now understand better why probiotics may also help in some people but not in others. The reason for it is the microbial environment, which varies greatly from person to person.”
Some of this research used germ-free mice; they start out without a microbiome, so the microbes that colonize these mice can be carefully controlled by the researchers. A colony consisting of twelve strains of common microbes was used in this report. But it's still not reflecting the true natural complexity of the microbiome. Yet, it also revealed how intricate that web of relationships can be. More work will have to be done before we can realize the therapeutic potential of microbes and probiotics, but this resarch has shown that beneficial microbes can be effective at promoting good health.
Sources: German Center for Infection Research, Cell Host & Microbe