Rare Genetic Variant Can Make People Susceptible to Bird Flu H7N9
Zoonosis is a serious health concern, as the COVID-19 pandemic has shown. Viruses that infect one species can acquire genetic mutations that enable them to infect other species. Researchers have been trying to learn more about exactly how that happens. If we can understand the mechanisms of transmission, we may eventually be able to prevent new zoonotic diseases from emerging.
Certain conditions seem to encourage diseases to jump the species barrier. For example, when humans live or work in close proximity to animals, there are lots of chances for a microbe to mingle with others from different hosts. In animal markets, workers may come into frequent contact with body fluids from animals that contain many different microorganisms. Microbes are good at exchanging genetic material with one another, so by incubating them all together, a zoonotic pathogen is more likely to emerge.
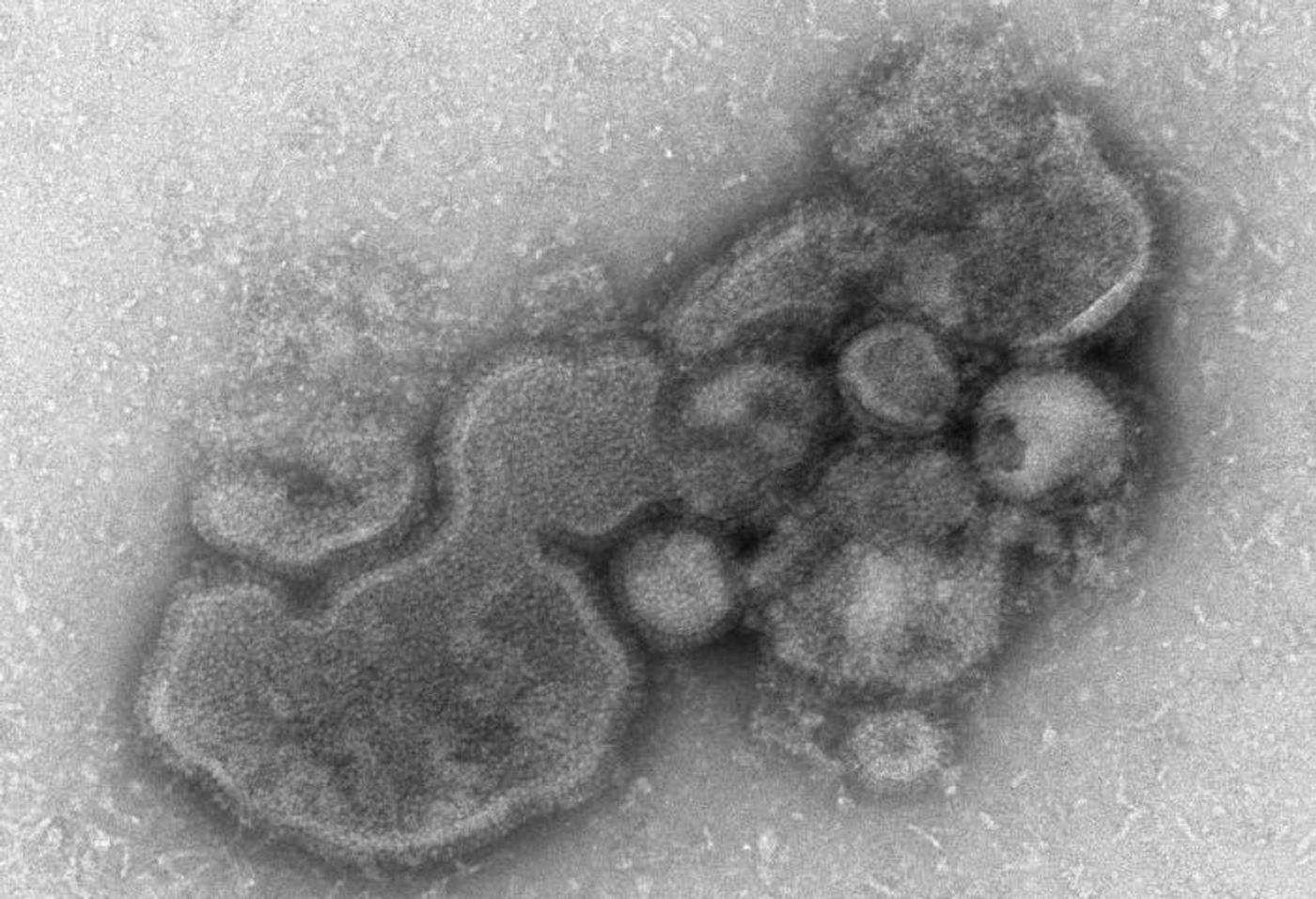
Though it is still unusual for avian influenza A virus (IAV) to infect people, when those viruses do enter a human host, they are dangerous; the mortality rate for IAV is over 30 percent. Scientists have not yet observed sustained transmission of IAV, which the World Health Organization defines as easy spread from one person to another. Viruses with an R0 that is higher than one are considered to have sustained transmission.
Reporting in Science, researchers theorized that without sustained transmission, host genes were likely playing a role in IAV infection in people. They performed whole genome sequencing on 220 Chinese people that had been infected with H7N9 between 2013 and 2017. The genomic data was compared to data from healthy individuals. The work revealed that these infections were strongly associated with small changes (single nucleotide variations) in the sequence of a gene called MX1.
MX1 encodes for a protein called myxovirus resistance protein A (MxA), which has been shown to have antiviral effects in a mouse model. Thus, people with certain variants in MX1 appear to be susceptible to H7N9 infection.
When the researchers introduced MxA variants into human cells, then exposed those cells to avian IAVs, including H7N9, most of the cells were not able to fight off the infection. Fourteen of seventeen MxA variants could not inhibit the virus. This study shows that MX1 plays a crucial role in host antiviral defense.
The study also suggested that when individuals carrying these MX1 variants become infected with avian flu, they could potentially act as a kind of reservoir in which mutations might occur that could potentially lead to sustained transmission of the virus. The researchers suggested that screening for MX1 variants in vulnerable populations may become a part of surveillance for emerging diseases.
Source: Science