Thermo Fisher Scientific Inc. (NYSE: TMO) is the world leader in serving science, with annual revenue of approximately $40 billion. Our Mission is to enable our customers to make the world healthier, cleaner and safer. Whether our customers are accelerating life sciences research, solving complex analytical challenges, increasing productivity in...
Rapid AST: A key weapon in the fight against AMR

Global consumption of the agents grew by 39% between 2000 and 2015 – and experts fear this will only get worse as the true scale of antibiotic prescribing during the COVID-19 pandemic becomes clear.
Of course, antibiotics are an essential tool in our medical armoury; we know that early antimicrobial treatment is associated with better outcomes in infections such as septic shock and bacterial meningitis. But we also know that the drugs are often used inappropriately.
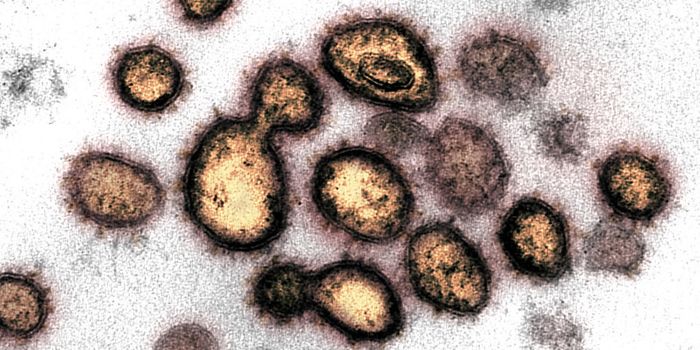
In sepsis, for example, clinicians often take a “just in case” approach to treatment. They prescribe broad-spectrum antibiotics while they wait for antibiotic susceptibility testing (AST) data to identify the pathogen, determine its susceptibility to agents, and define the minimum inhibitory concentration (MIC) needed to inhibit growth.
It is a process that can, with traditional methods, take up to 72 hours. Delaying the administration of targeted, personalised treatment thwarts the efforts of AMR stewardship programmes, which aim to preserve antimicrobials by ensuring the right person gets the right treatment, at the right time
Rapid, robust results
In a world where lost time can mean lost lives, faster diagnostics are essential, and the scientific community has been calling for ways to shorten the time from sample collection to lab result for decades.
Faster AST would enable more timely clinical decisions, contributing to better patent outcomes, and helping to stem the AMR tide, they say.
Studies have shown that returning an AST result 24 hours earlier than standard time frames could deliver per patient cost savings of between $2,500 and $20,000, and cut mortality rates by a staggering 40%.
However, advances have been elusive, as most approaches have focused primarily on the time to result. Now evolutions in technology are allowing for a more method-focused view that ensures results are not only rapid, but reliable, informative, and actionable.
Method-based approach
The ISO standard for AST, ISO 20776-1, describes a broth micro-dilution reference method and highlights the importance of MIC. It is a value that will differ greatly depending on multiple factors, including the strain and clinical case – and for some antibiotics and bacteria, MIC is the only reliable phenotypic method for assessing drug sensitivity.
Truly clinically valuable rapid AST, then, will not only return quicker results, but it will also return the all-important MIC value, reflecting the gold standard method as closely as possible.
The ASTar® system, developed by Q-linea, distributed by Thermo Fisher Scientific, and based on the ISO 2776-1 rapid AST broth microdilution method, does just that.
Using cutting edge advanced imaging techniques and algorithms to monitor bacterial growth during incubation, without the need for the lengthy subculture step, the system, it can deliver accurate, actionable results in just six hours.
Crucially, it produces the MIC results clinicians need to deliver personalized antibiotic treatment – and continue their fight against AMR.
Find out more: https://assets.thermofisher.com/TFS-Assets/MBD/brochures/Smartnote-Rapid-AST-LT2678A.pdf
Product is CE marked but not 510(k)-cleared and not available for sale in the U.S. Availability of product in each country depends on local regulatory marketing authorization status.
Q-linea AB is the legal manufacturer of ASTar®. ASTar® is a trademark of Q-linea AB. The ASTar system is distributed globally by Thermo Fisher Scientific.