Researchers at NYU Langone Medical Center found that helminth infection in the gut triggers immune responses that alter the gut microbiome, mitigating inflammatory bowel disease (IBD).
These results lend further support to the hygiene hypothesis. This hypothesis states that the absence of parasites - due to our exceptionally hygienic lifestyle - leaves the immune system highly responsive to changes in the gut. Supporters of the hygiene hypothesis think that a lack of exposure to antigens (microorganisms, parasites, etc.) early in life prevents the immune system from developing properly. This, in turn, affects how well the body establishes
immune tolerance - making people susceptible to autoimmune disease.
In fact, there is indeed a correlation between the low incidence of parasites in the developed world and high rates of inflammatory bowel diseases like ulcerative colitis and Crohn’s disease. According to study author P’ng Loke, “our findings are among the first to link parasites and bacteria to the origin of IBD, supporting the hygiene hypothesis”.
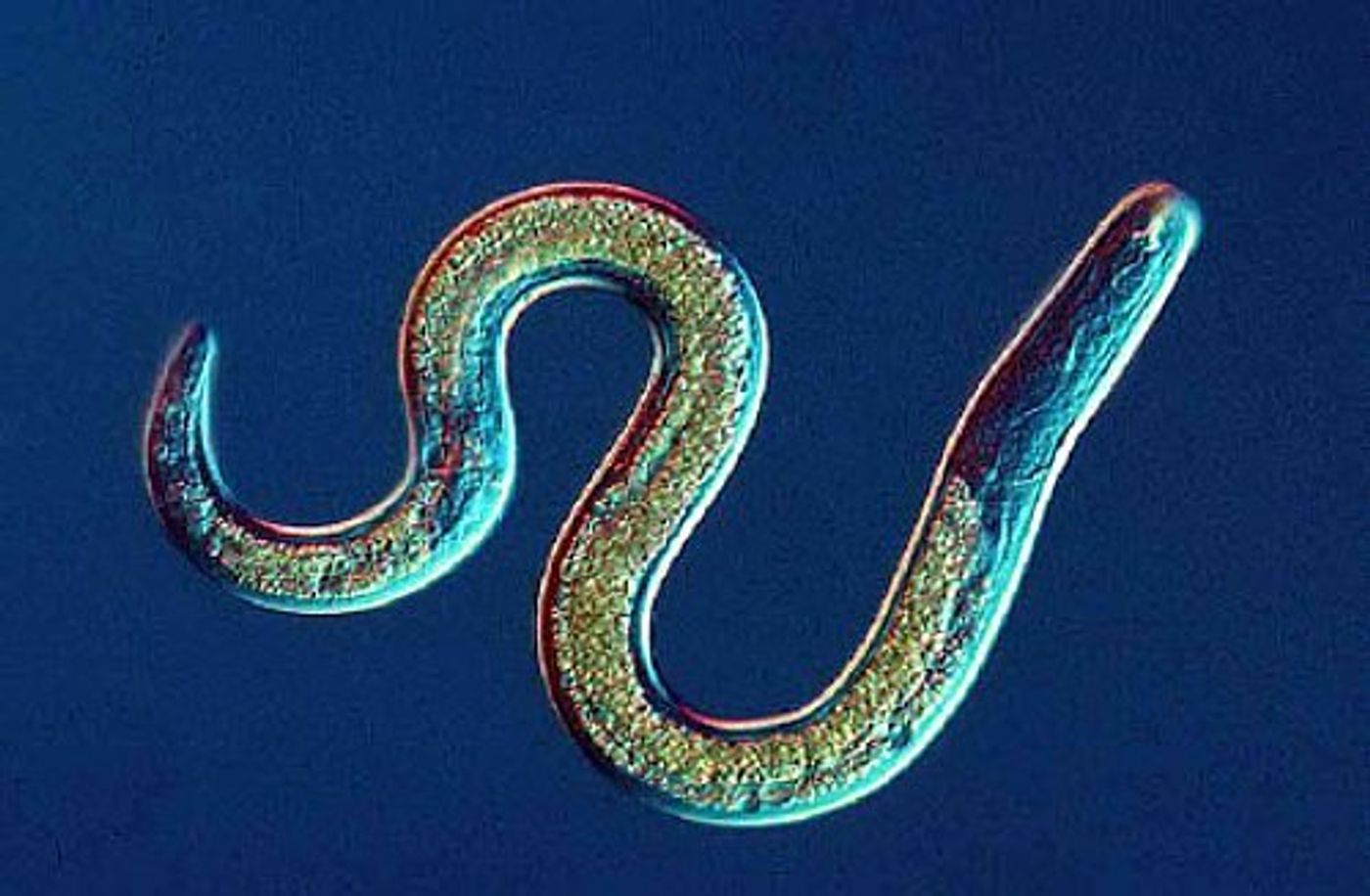
In their study, published in Science, the NYU researchers infected mice with helminths (intestinal worms) and characterized changes in the gut microbiome. What they saw was striking - nearly a thousand-fold decrease in bacteria belonging to the genus Bacteroides, and these bacteria are associated with IBD. The helminth infection also increased the number of species from the class Clostridia. These bacteria are associated with anti-inflammatory effects.
So, how do intestinal worms prevent IBD? The researchers think that the immune response to the worms produces an environment that is favorable to Clostridia, allowing these cells to grow. At the same time, this environment may be inhospitable to Bacteroides.
The study also reports that people living in rural Malaysia have relatively high rates of worm infection, low rates of IBD, and more Clostridia than Bacteroides in their guts. When these people were treated for the worm infections, they developed more Bacteroides than Clostridia in their microbiomes.
These findings could be the basis for future IBD treatments that involve treating patients with immune-stimulating drugs, mimicking the response to a worm infection.
Sources: Science,
Science Daily, Wikipedia