It’s been known for some time that the symbiotic relationship between a microbiome and the host requires a fine balance in order to maintain health. A multitude of external factors can influence the microbiome, including diet, genetics, and the environment. Problems with the interconnection of mammalian host and microbiota are associated with the pathogenesis of multiple diseases such as irritable bowel syndrome, nonalcoholic fatty liver disease, inflammatory bowel disease, and gastrointestinal malignancy.
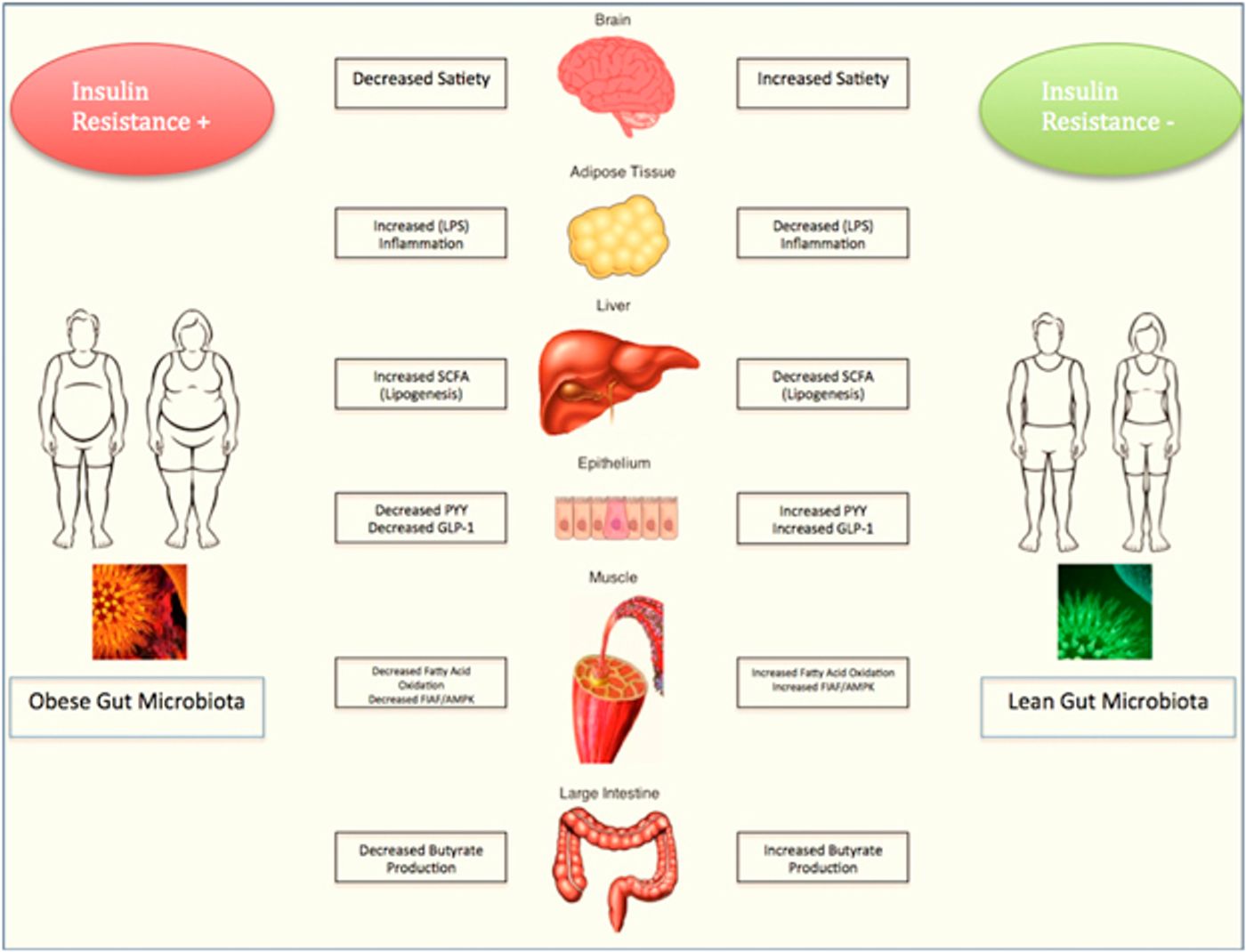
While insulin resistance, obesity, and metabolic syndrome have also been correlated with changes in the gut microbiome, no causal link had been established until the release of this
new report in Nature. Researchers have been working on demonstrating causation for some time. Gerald I. Shulman, M.D., the George R. Cowgill Professor of Medicine at Yale University and a Howard Hughes Medical Institute investigator, has previously observed that a short-chain fatty acid, acetate, provoked the secretion of insulin in rodent models. To investigate acetate's exact role, Shulman and his team conducted a series of experiments in obese rodents.
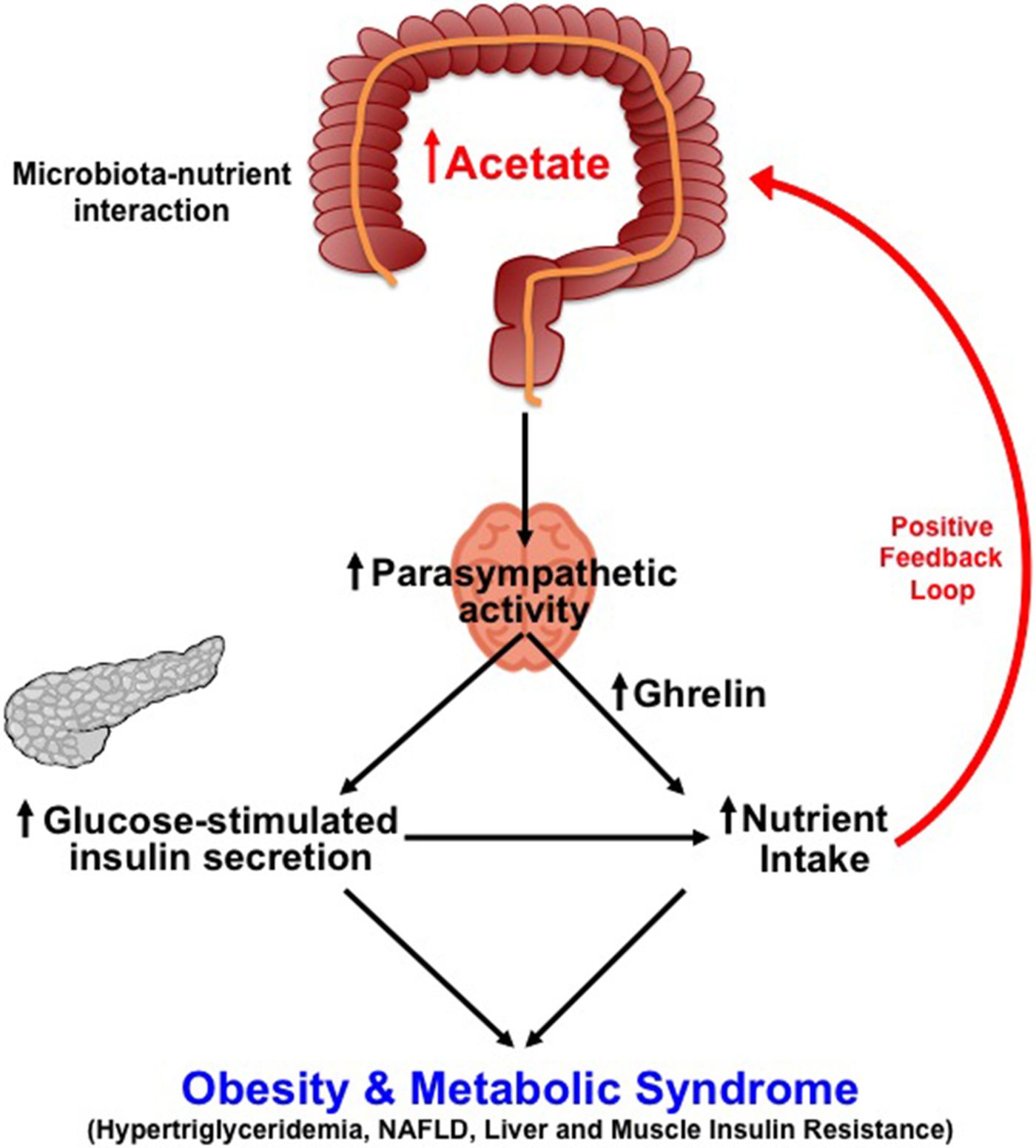
They measured and found higher amounts of acetate in animals that had eaten a diet high in fats. Then they observed that when the animals were given acetate, whether by infusion or by direct injection of acetate into the brain, insulin was then released by the beta cells of the pancreas. "Acetate stimulates beta cells to secrete more insulin in response to glucose through a centrally mediated mechanism," explains Shulman. "It also stimulates secretion of the hormones gastrin and ghrelin, which lead to increased food intake."
In order to prove a cause and effect relationship, the scientists performed a fecal transplant from one group of animals to another, and showed that there were alterations in acetate and insulin levels as well as changes in gut microbiota.
"Taken together these experiments demonstrate a causal link between alterations in the gut microbiota in response to changes in the diet and increased acetate production," says Shulman. The increased acetate in turn leads to increased food intake, setting off a positive feedback loop that drives obesity and insulin resistance, he explained.
The study goes on to suggest that the feedback loop could be important during evolution. It would have prompted animals to gorge and build up body mass when they were fortuitous enough to encounter calorically dense food during times of food scarcity.
"Alterations in the gut microbiota are associated with obesity and the metabolic syndrome in both humans and rodents," Shulman continues. "In this study we provide a novel mechanism to explain this biological phenomenon in rodents, and we are now examining whether this mechanism translates to humans."
Sources:
Nature: Parekh et al,
Nature: Perry et al,
Yale University