In a new issue, Science Translational Medicine presents two related papers that demonstrate variations in the gut microbiomes of infants based on mode of birth, diet and antibiotics. Gut microbiota, or the population of microbes that a human carries in their intestinal tract, goes through a lot of changes in the first couple years of life, then it tends to stabilize. What happens during early development appears to have a lifelong impact on health.
“The take-home message is that not allowing the immune system to develop to its maximum potential … is not a good thing,” said Dr. Ramnik Xavier, senior author of one of the
studies and chief of the gastrointestinal unit at Massachusetts General Hospital. “You need the microbiome to develop normally and educate the immune system in the gut to respond to these microbes and also prevent other harmful bugs from getting in.”
Xavier's
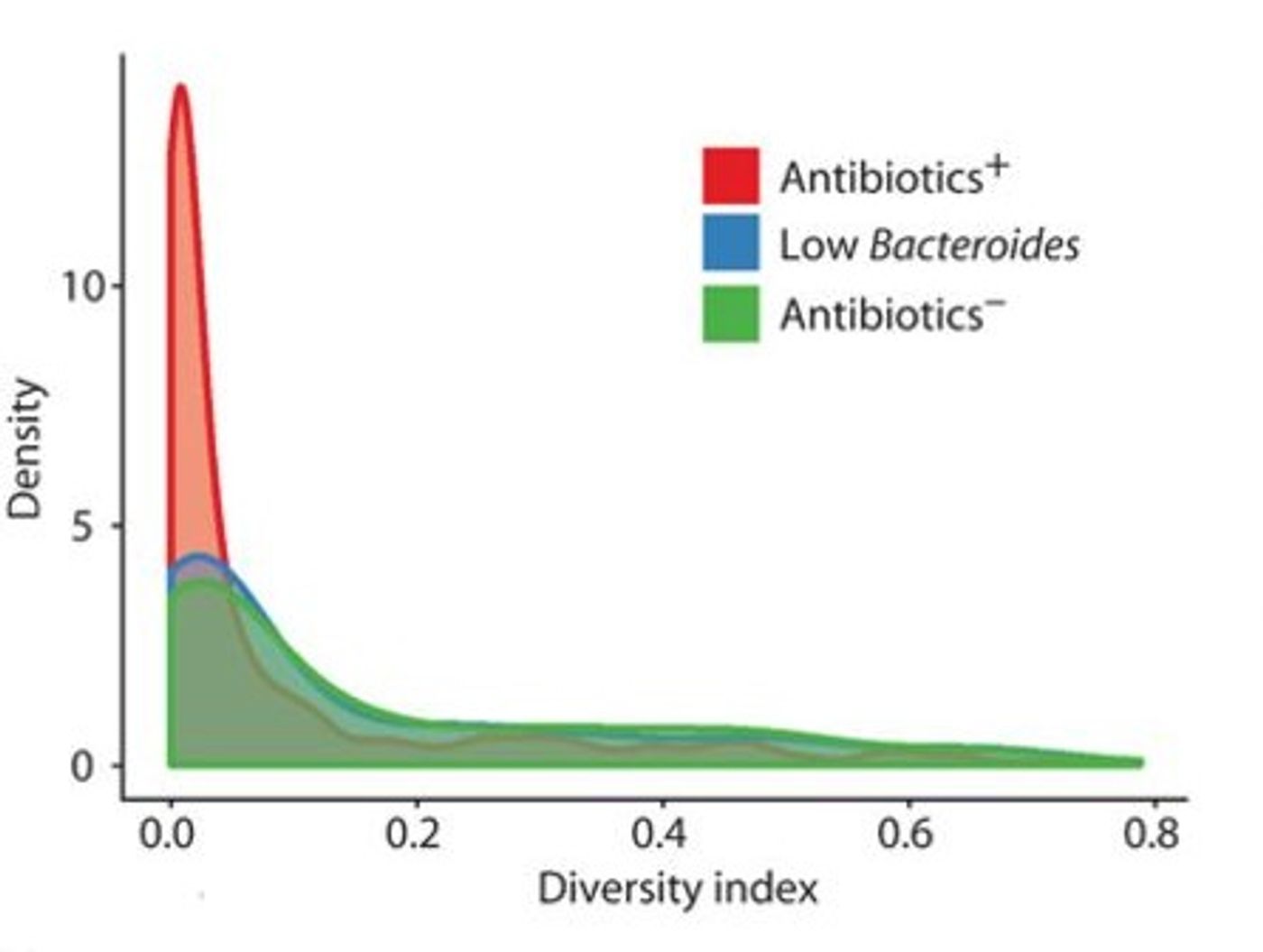
report monitored 39 kids over three years, with half receiving antibiotics and the other half getting none, with around 28 samples taken throughout that period. Many babies in the study had received multiple rounds of treatment, some up to 15 treatments.
Children treated with antibiotics had microbiomes that were unstable and less diverse compared to those who had not. In some children, the flora was not only dominated by a single species, but was primarily composed of only a single strain within that species. The researchers also saw short-term changes in bacterial composition between consecutive samples from children given antibiotics. The research team observed an increased amount of antibiotic resistance genes after drug treatment, with simultaneous increases in bacteria that are likely to harbor these genes. That could be detrimental to the efficacy of such treatments in the child’s future.
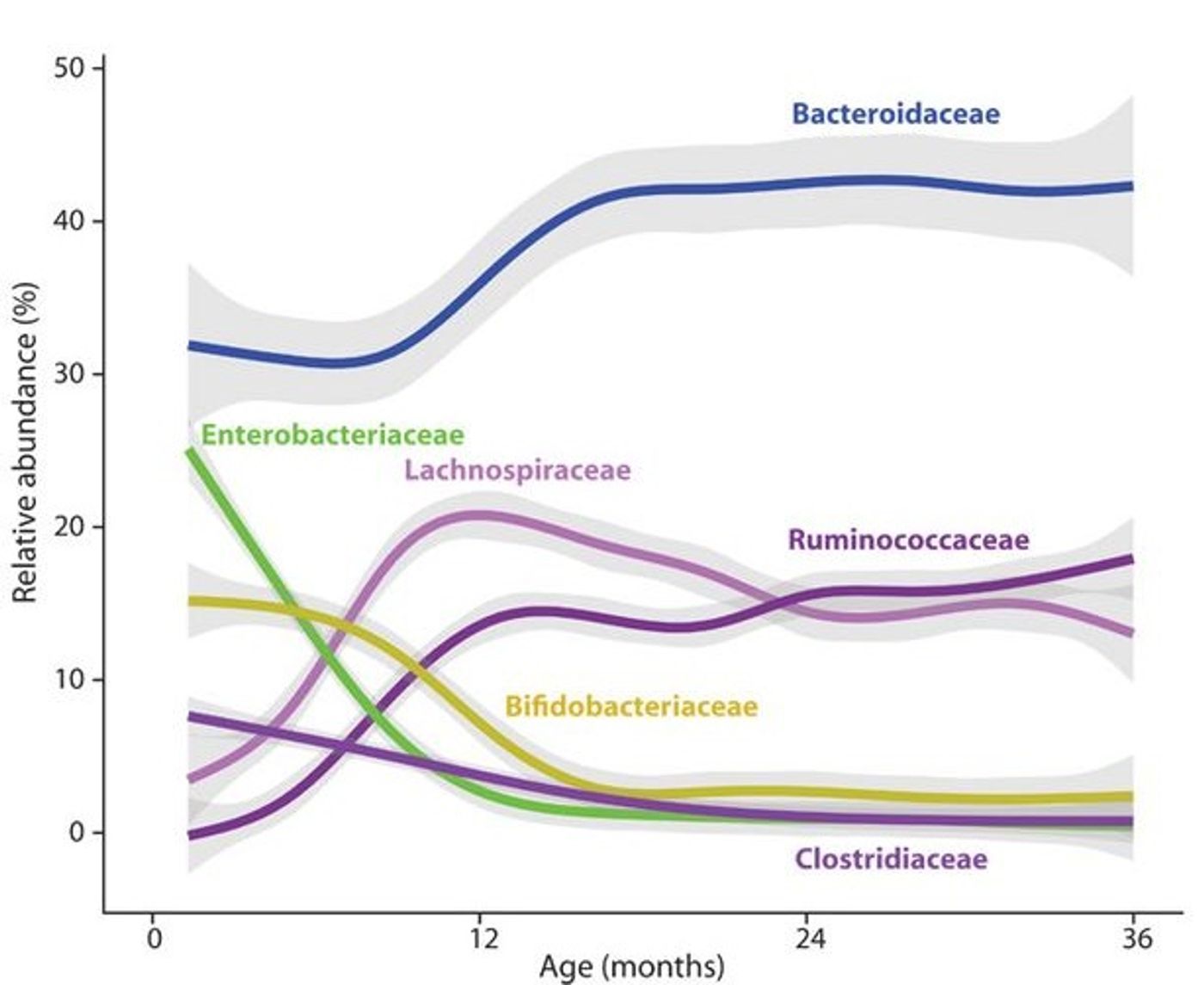
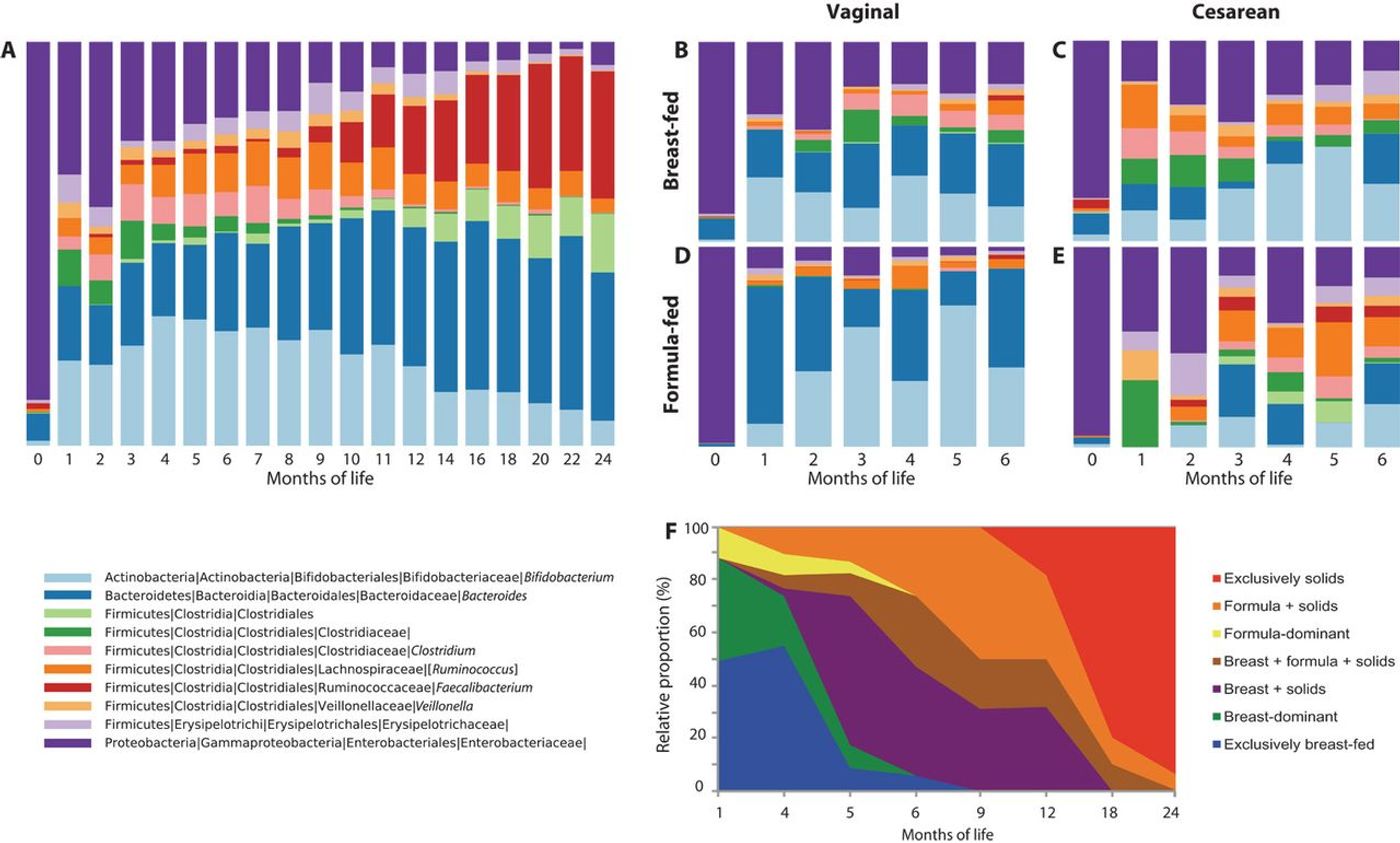
This same study also showed a difference in the microbiota of children who had been delivered by cesarean section versus vaginally. One species of bacteria that dominated the intestinal flora of most children born vaginally, Bacteroides, was completely absent from the guts of children born by cesarean until they were six to 18 months old.
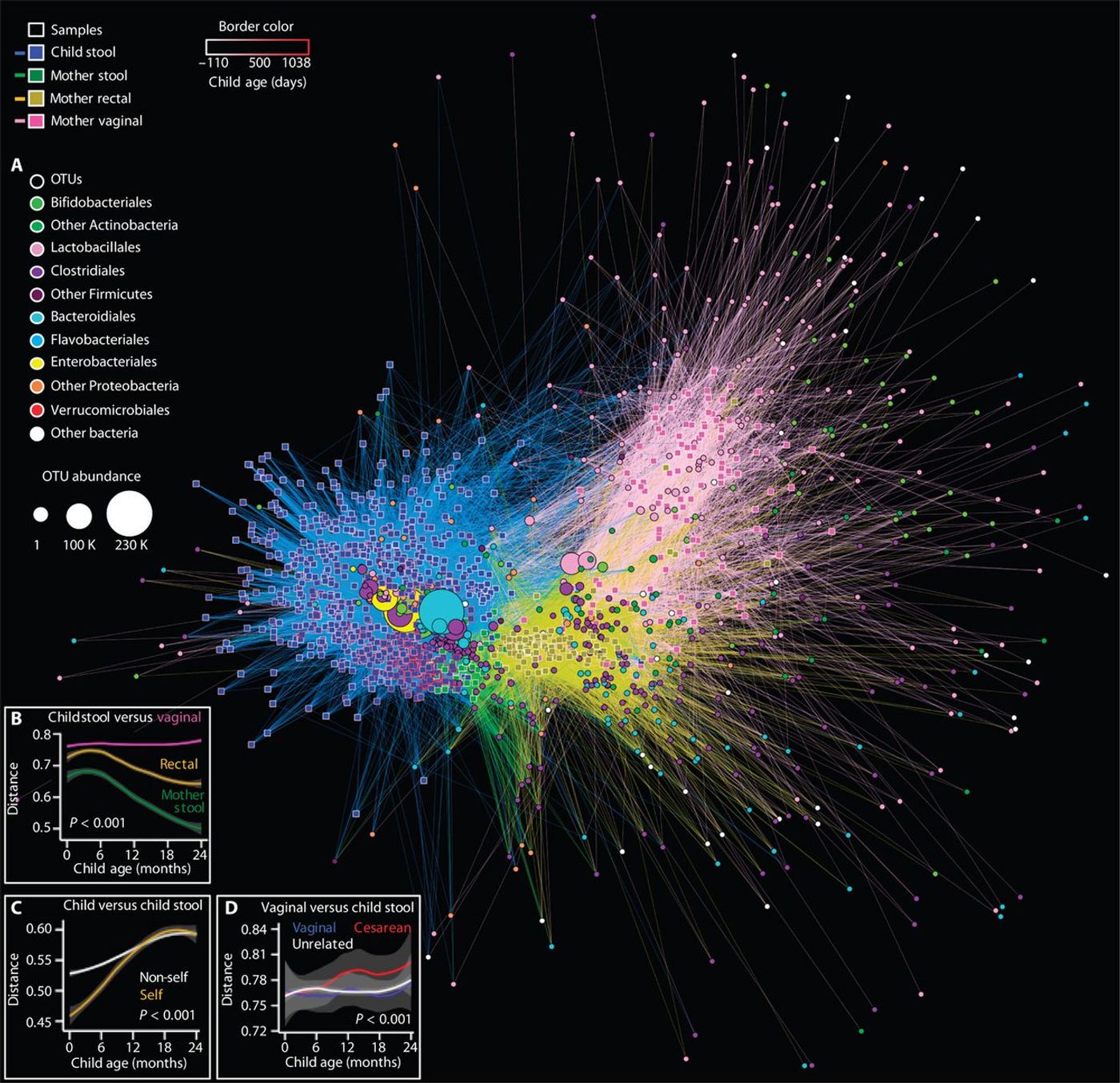
A second
study found similar results when they looked at a group of 43 children over two years. They found that exposure to antibiotics, cesarean section, and formula feeding perturbed the microbiome, altering its diversity, delaying its maturation and changing the establishment of maternal bacteria. An author of this study, Dr. Martin Blaser, a professor at New York University, commented, “there’s still a period of time when the microbiome is not maturing at the same rate as the baby.”
While one cannot establish cause and effect relationships from these studies, it has become apparent that delivery mode, diet and drug treatments are important to evaluate on a case-by-case basis because of the potential long term effects on health of these decisions.
The video below is a pitch for an indiegogo documentary on the subject of infant microbiota and antibiotics, but it contains some informative commentary on this topic from experts in the field.
Sources:
STAT News,
Science Translational Medicine Yassour et al,
Science Translational Medicine Bokulich et al