Vitamin A has roles in the production of light receptors for the eye as well as the normal development of the fetus and placenta. It is critical for mammals. For more on Vitamin A, watch the video below.
"Being able to visualize this protein, and understand how it works to move Vitamin A, is a really fantastic leap," commented one co-authors of the
Science paper, David J. Weber, PhD, Professor of Biochemistry and Molecular Biology at UM SOM. "And there is so much more we can do with this technique. It's exciting."
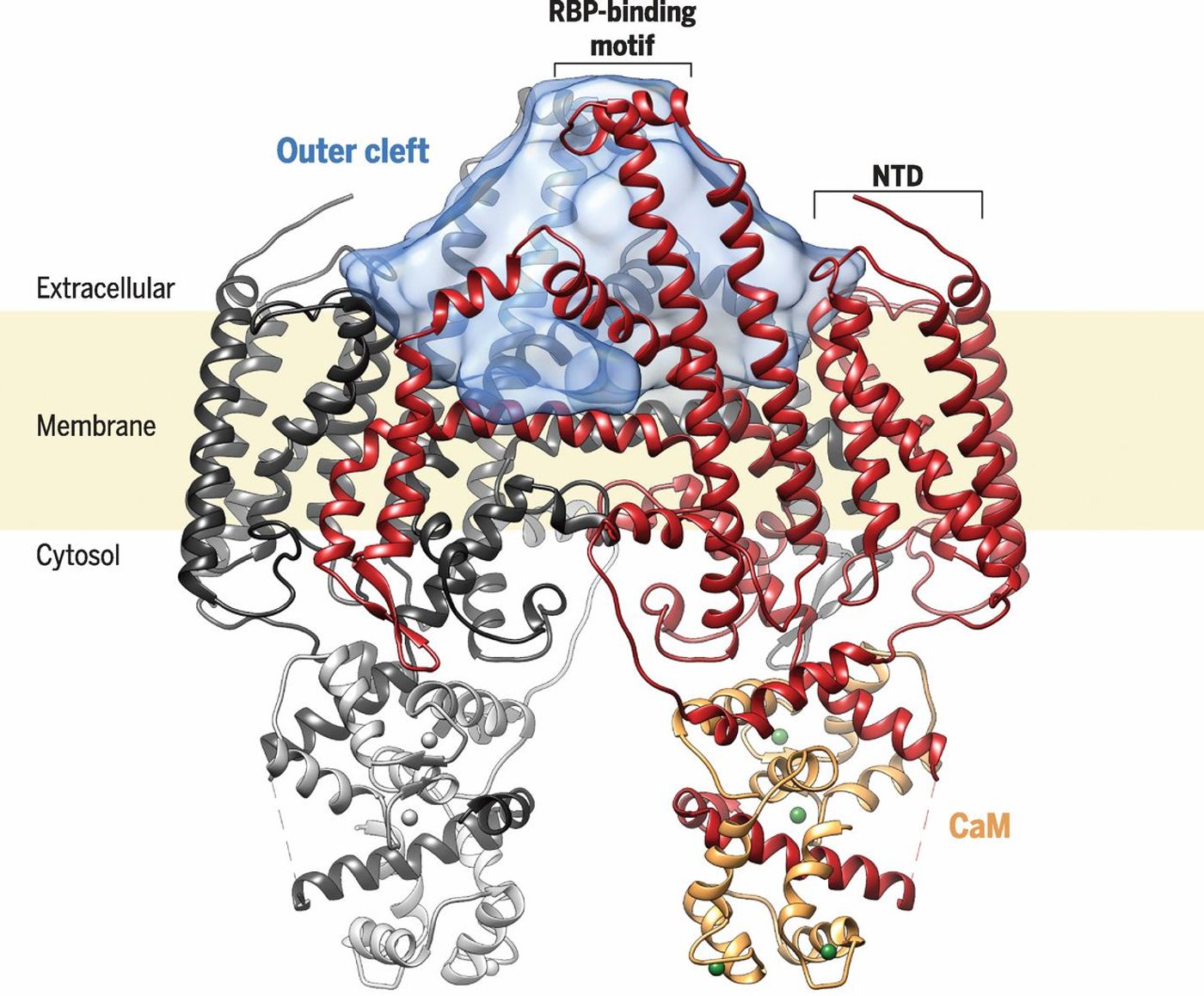
The work was the culmination of effort by a team of researchers led by Filippo Mancia, PhD, Assistant Professor of Physiology and Cellular Biophysics at Columbia University Medical Center, and has been published in Science. An electron microscope was combined with incredible speed to capture images of the protein, which had some interesting characteristics.
"We can now get near atomic resolution because the new camera is much faster and allows us to take a movie of the molecules," explained Oliver Clarke, PhD, an associate research scientist in the lab of Wayne Hendrickson at Columbia University Medical Center. "Even under the electron microscope, the molecules are moving around by a tiny amount, but when you take a picture of something moving, it comes out blurry. With such a movie, we can align the frames of the movie to generate a sharper ima
Using about 70,000 individual pictures of STRA6, the research team was able to create a 3D map of the protein, from which a precise atomic model was made.
The mechanism of Vitamin A transport by STRA6 was revealed by this study. It uses an intermediary protein rather than utilizing a direct interaction to move Vitamin A. By Clark’s estimation, that makes STRA6 "a bit of a freak." It may serve as a way to prevent cells from getting too much Vitamin A.
"Vitamin A is actually somewhat toxic," said another co-author, Chiara Manzini of George Washington University. "Trapping vitamin A inside the membrane may keep control of the amount inside the cell."
While additional confirmation will be required, this could be a way to explain some other mechanisms of transport.
"This collaboration among research institutions has yielded fascinating insight into the cellular pathway of vitamin A," commentedd UM SOM Dean E. Albert Reece, MD, PhD, MBA. "When scientists work together like this, great accomplishments occur. The technique developed here will clearly reap future discoveries in other domains as well."
Sources:
AAAS/Eurekalert via UM SOM,
Science