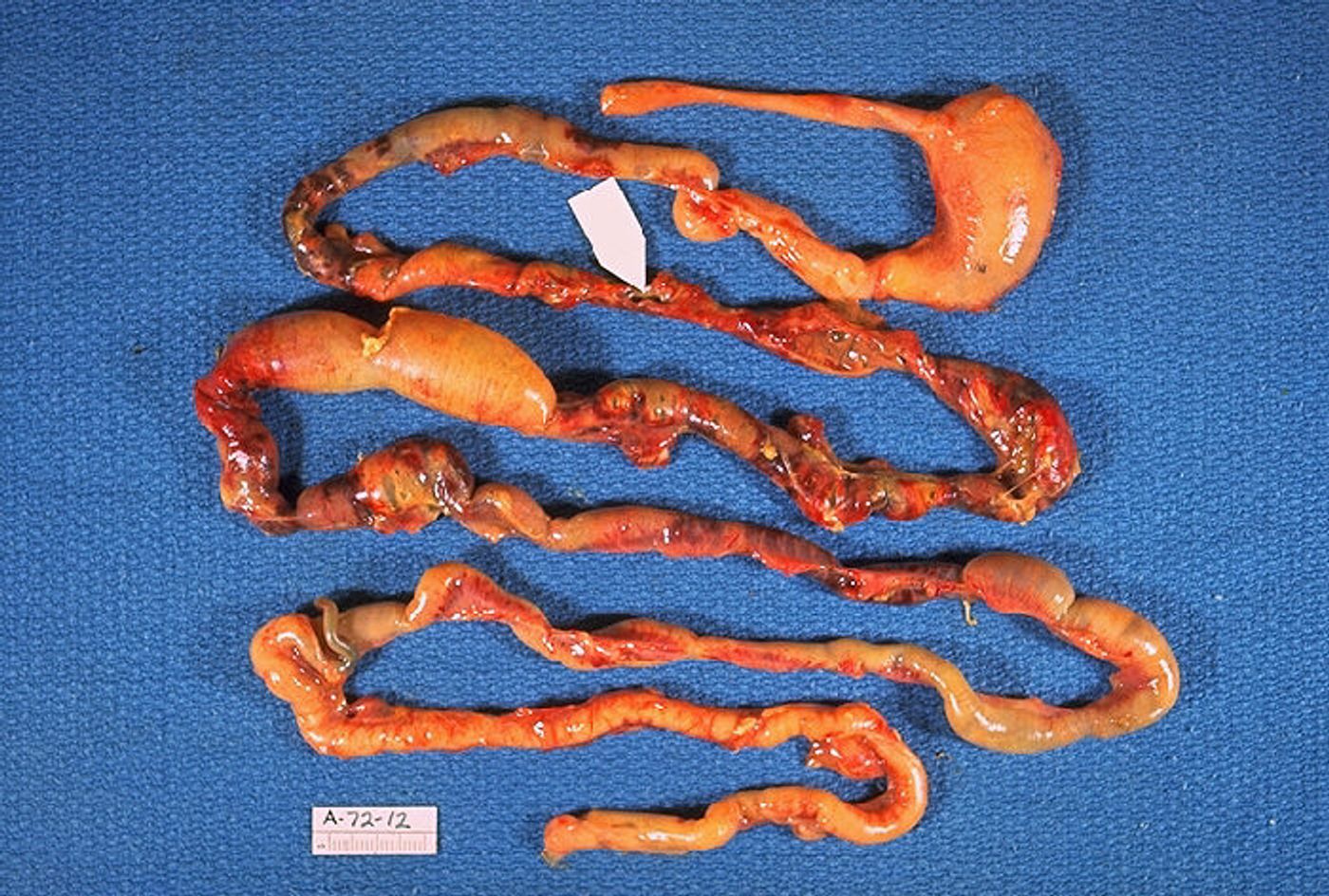
Necrotizing enterocolitis is a nasty ailment. It’s a condition common in premature infants in which the intestines become inflamed and necrotic, sometimes leading to death. Often, bacteria invade the damaged bowel wall, further complicating things.
For the clinically-inclined, necrotizing enterocolitis (NEC) is dividied into Bell’s three stages of disease. The first, stage 1, encompasses “suspected disease”. Stage 1 symptoms include temperature instability, abdominal distension, and bloody stools. Stage 2 is defined as “definite disease”, and symptoms include metabolic acidosis and thrombocytopenia. Finally, stage 3 is “advanced disease”, with symptoms ranging from severe abdominal distension to metabolic and respiratory acidosis.
Early on, investigators thought that NEC was caused primarily by ischemic injury in premature infants (NEC occurs in up to 5% of ICU admission) - for whatever reason, the intestines did not receive adequate blood flow and died. Various other risk factors are now recognized. These include formula feeding and abnormal intestinal microbiota. If you’re reading this article for the microbiology content, here we go.
As with many things these days, researchers recognize a connection between the intestinal microbiome and NEC. Such NEC-inducing changes to the microbiome include a decrease in commensals and an increase in pathogens; these changes are associated with the use of antibiotics and acid-suppressing medications.
We still have much to learn about the neonatal microbiome. (It’s still in its infancy, you could say. See what I did there?) What’s clear is that the gut undergoes enormous changes after birth; it must cultivate a healthy population of commensal bacteria (to digest food and produce vitamins, for starters). Among the first residents of the gut are Enterobacteriae, Enterococci, and Staphylococci.
The big question is, do infants at risk for developing NEC have different microbiomes than other infants? Several studies report that changes to the microbiome can be observed anywhere from 72 hours to 3 weeks before NEC develops. A culture-based study from some 20 years ago associated NEC with increases in the abundance of E. coli and Enterobacter cloacae. More recently, sequencing-based methods report decreases in the abundance of Bifidobacteria and Klebsiella species before NEC was diagnosed. It’s worth pointing out that it’s not clear whether these changes to the microbiome cause NEC or whether some other, unidentified change causes NEC and also disrupts the microbiome.
Whether microbes are fully to blame for NEC, it’s clear they can complicate things. NEC is associated with bloodstream infections, typically with Gram-negative bacteria like E. coli and Klebsiella. Again, it’s not clear whether NEC weakens the intestinal barrier, leading to systemic infection, or whether the systemic infection occurs upstream of NEC.
So, what can be done about NEC? We can harness the power of the microbiome, of course! It seems that many news headlines tout probiotics as a panacea - for the record, I’m not convinced. There appears to be good data, however, for using probiotic bacteria like
Bifidobacteria and
Lactobacillus to prevent and treat NEC. After all, probiotics are reported to attenuate NF-κB activation (reducing inflammation), increase the expression of cytoprotective genes, and decrease cell death.
A study from 2010 showed that 20 out of 24 randomized controlled trials demonstrated the efficacy of probiotics for reducing NEC severity and overall mortality. Groups in Canada and Germany also report a drop in cases of NEC in hospitals that regularly use probiotic therapy.
Sources: Wikipedia,
Drugs,
The Journal of Pediatrics,
Cochrane Library,
Pediatric Research