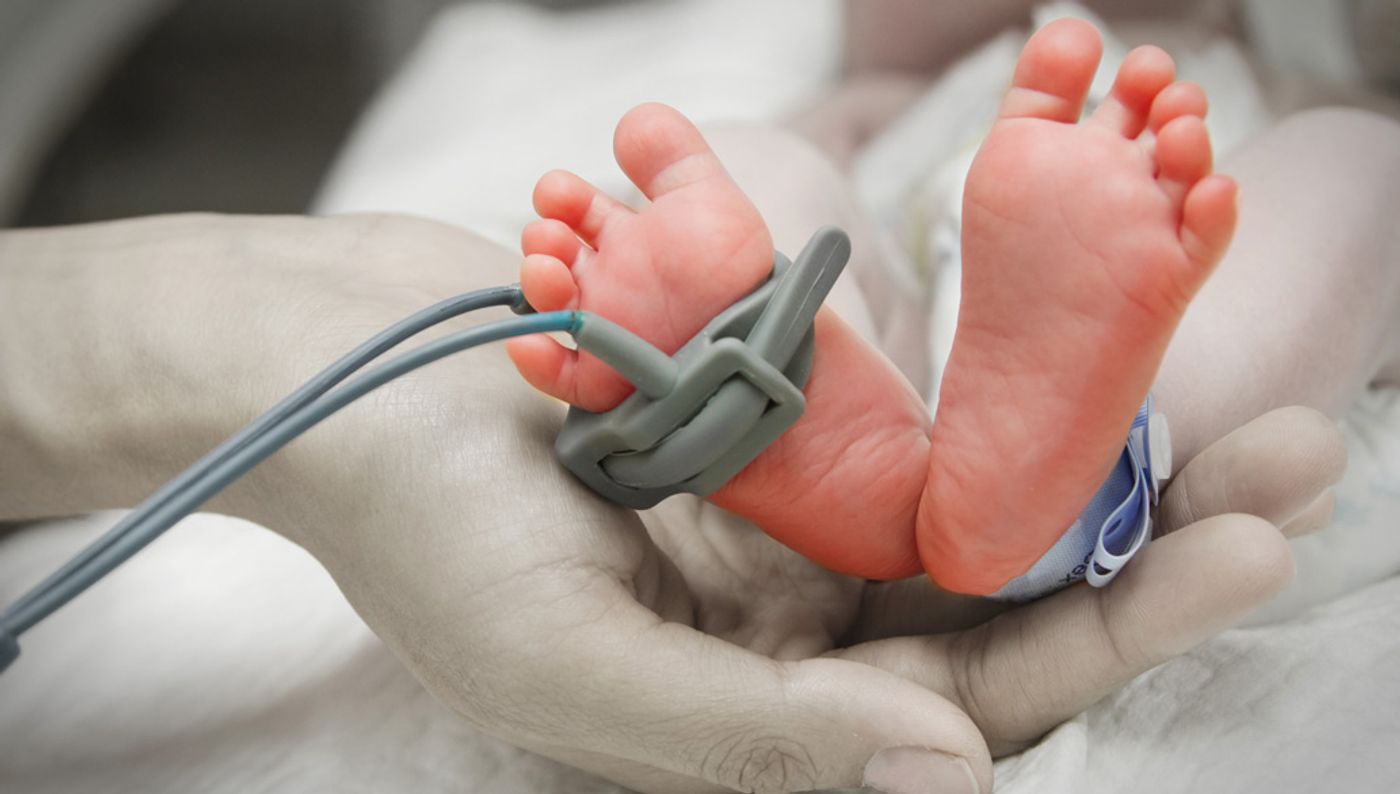
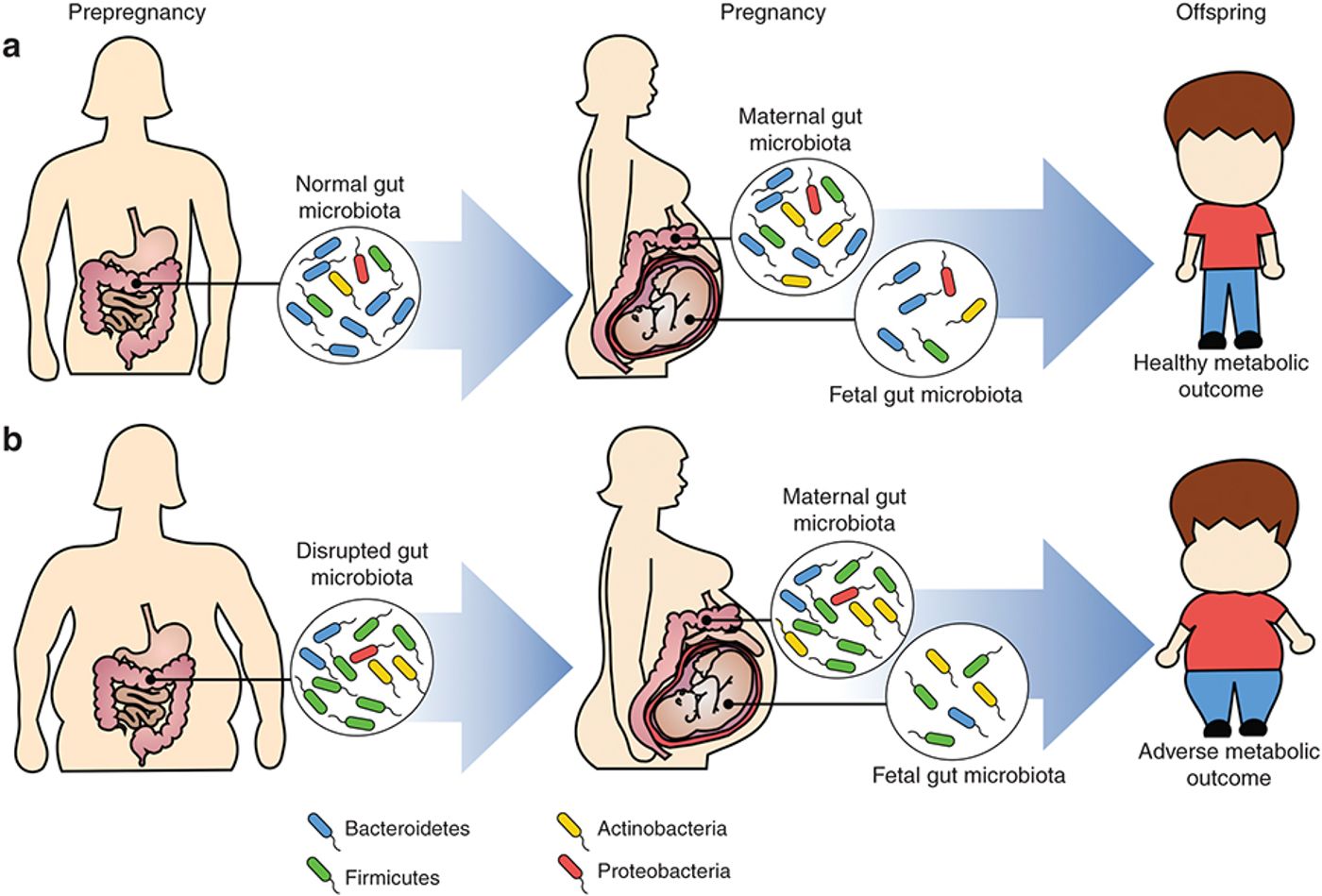
The first month of a newborn’s life is so critical to a lifetime of health that this period has a name: neonate. In a new collaboration between the University of California San Francisco and the Henry Ford Health System, scientists made clear just how important a healthy microbiome is for neonates and the future of their allergy and asthma status.
"Currently, children are typically six or seven years old when they are diagnosed with asthma, which has no cure and has to be managed through medication,” explained co-senior author Susan Lynch, PhD from UCSF. “If we are to prevent disease development, we need to intervene early.”
The key to detecting asthma in individuals as neonates, the current study’s findings suggest, is through the neonatal microbiome.
From the Henry Ford Health System, researchers took stool samples from a local study focusing on the role of environmental factors in allergy and asthma, over one hundred samples from neonates. Using the high-throughput genetic analyses technology available at UCSF, the researchers gained detailed insight into the microbiome composition of each individual stool sample, and they collected their findings into three distinct groups determined by the bacterial and fungal diversity evident in each neonate’s microbiome.
"It seems that the microbial communities within the body could be the keystone to understanding this and a number of different immune diseases,” said co-senior author Christine Cole Johnson, PhD, MPH from the Henry Ford Health System.
According to both two-year and four-year follow-ups with the neonates whose stool samples were used in the initial study, the researchers saw a significant increase in asthma and allergy rates for the individuals in the smallest grouping of microbial composition, and this was also the group whose stool samples were missing the most “normal” gut bacteria and had abnormally high levels of specific resident fungi.
The ratio of neonates who were at an increased risk for asthma and allergies later in life - specifically three times as likely to develop allergic reactions at age two and asthma at age four - is representative of the general population: just under ten percent of the neonates were in the high-risk group.
Next, the team went one step further. By studying the metabolic byproducts of the microbes from the stool samples, they were able to understand at a molecular level the connection between an unhealthy microbiome lacking certain species and the elevated risk for asthma and allergies.
In a healthy microbiome, containing all of the “normal” bacteria that researchers see in the guts of healthy individuals, researchers saw a large population of anti-inflammatory lipids that nourished a healthy population of regulatory T cells, lymphocytes of the immune system that regulate the immune response, preventing autoimmune reactions like those present in asthma and allergies.
However, in an unhealthy microbiome such as those in ten percent of neonates in the study, anti-inflammatory lipids were absent. Instead, the microbiome byproducts of these samples had fat molecules known to be associated with asthma, 12, 13 DIHOME for example, which reduced the production of regulatory T cells. Researchers believe that the loss of a healthy regulatory T cell population is what causes the immune system to overreact, or become hyperactive, later in life, causing asthma and allergies.
"Asthma has doubled in prevalence in modern ‘westernized’ societies about every 20 years for the past 60 or 70 years, so an effective strategy for prevention is becoming an urgent need for public health," said Homer Boushey, MD, a UCSF team member. "By focusing on the differences in microbial functions -- in their metabolic products and their effects on immune function -- this kind of study helps define the pathway we'll need to follow to prevent this disease."
Both UCSF and Henry Ford Health System researchers from the study believe that the difference in metabolites between what they consider a healthy microbiome versus an unhealthy microbiome stems from the aid the microbiota provide in digesting dietary molecules, like fats.
Certain factors like breastfeeding, vaginal births, and even having dogs in the house during the neonatal period and up to one year after birth have been shown to have a protective effect on the health of newborns, all of which stem from contributing to a healthy microbiome.
Technology may make events like breast-feeding or vaginal births an option rather than a requirement, but the effects of these changes from centuries of individuals being subjected to the same treatment during and after birth are visible in the changing composition of the microbiome and subsequent impact on allergies and asthma. After all, the bacteria in the gut evolved over time just as humans did. How can we not expect repercussions when they’re suddenly removed from developmental processes?
Source:
University of California San Francisco,
Nature Medicine