Electron microscopy is a powerful tool that produces images of things magnified up to ten million times, but the images it produces are monochromatic. New research from University of California, San Diego (UCSD) has changed all that.
Reporting in Cell Chemical Biology, the scientists report a major advancement. Mark Ellisman and the late Roger Tsien have led the development of multicolor electron microscopy. Tsien won the 2008 Chemistry Nobel Prize for the development of GFP into a research tool, but unfortunately died in August 2016. The video below features a nice description of the work.
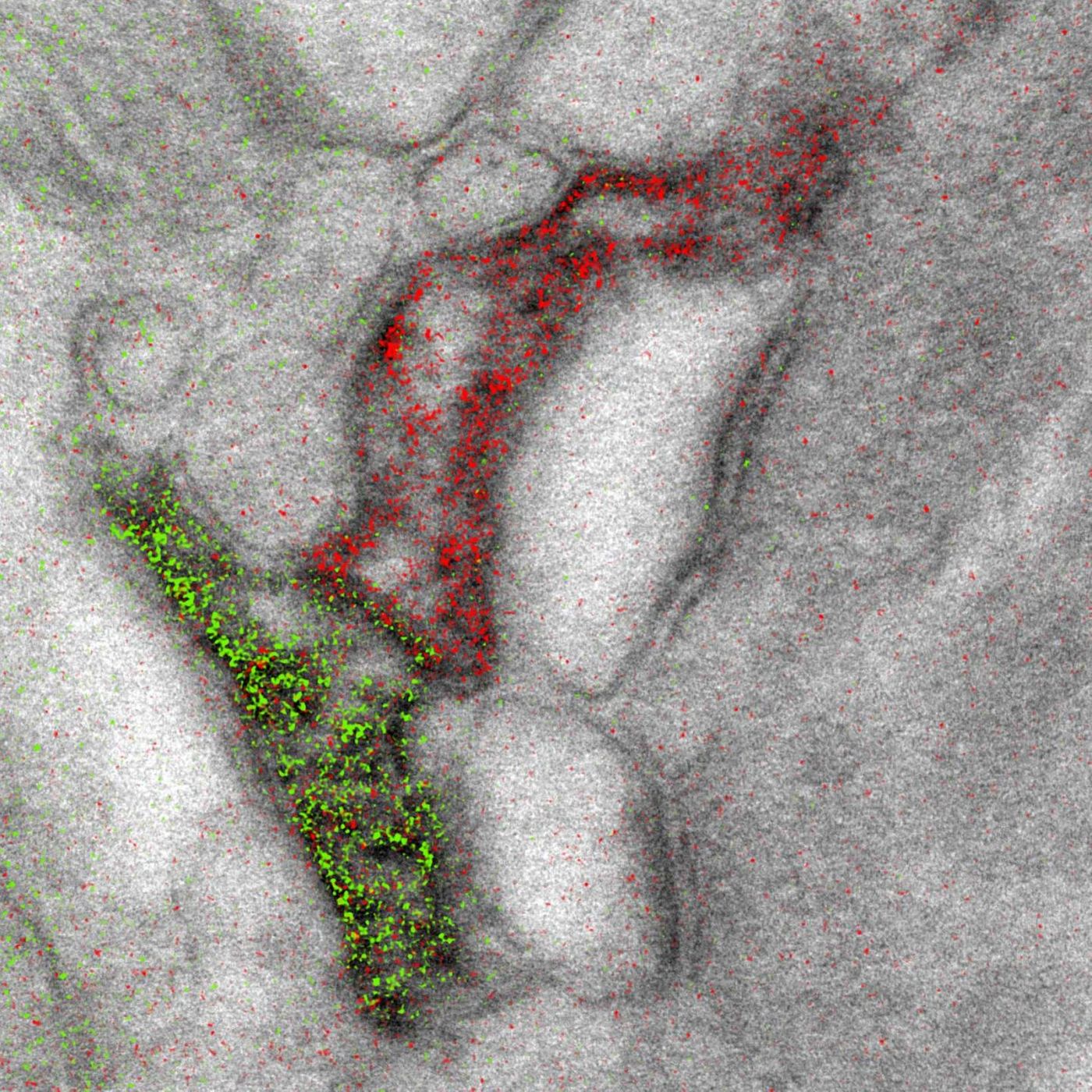
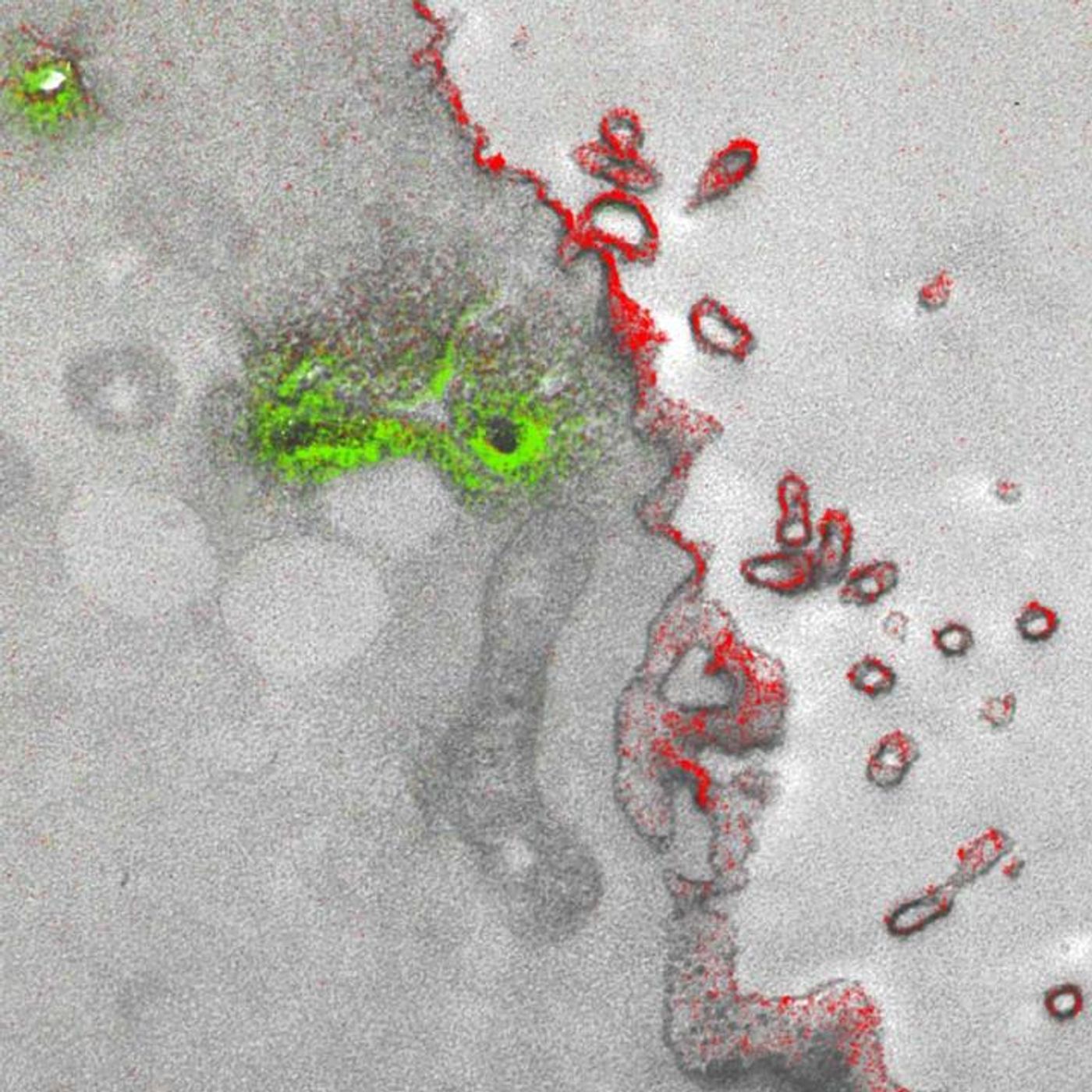
The new technique took almost 15 years to perfect. It allows up to three colors – green, red and yellow – to be used at once in an image. The microscope has a detector for the capture of electrons emanating from metal ions painted over the specimen, and the energy loss registers as a color. The ionized metals create an elemental map, which are overlaid with the microscopy image to create a multicolor image with incredible spatial resolution.
"It's a bit like when you first see a color photograph after having only known black and white -- for the last 50 years or so, we've been so used to monochrome electron micrographs that it's now hard to imagine that we could go back," commented the first author of the work, Stephen Adams, a UCSD chemist. "This method has many potential applications in biology; in the paper, we demonstrate how it can distinguish cellular compartments or track proteins and tag cells."
The investigators needed a special metal to achieve their goals; it had to be stable enough to handle the application without deteriorating or otherwise, it would blur the image, and it had to have a distinctive energy loss. The lanthanide family of metals was utilized, lanthanum (La), cerium (Ce), and praseodymium (Pr). The emitting electrons have to be deposited on to the specimen on the scope sequentially, a major hurdle the researchers had to overcome.
"One challenge that kept us from publishing this much earlier, because we had the chemistry and we had an instrument that worked about 4 years ago, was we needed a way to deposit the metal compounds sequentially," explained co-senior author Mark Ellisman, the Director of the National Center for Microscopy and Imaging Research at UCSD. "We spent an awful lot of time trying to figure out how to deposit one of the lanthanides and then clear it so that it didn't react when we deposited a second signal on the first site."
The researchers have demonstrated the feasibility of their technique by visualizing two brain cells connected in a synapse, and peptides moving through a cell membrane. This publication was also one of the last that Tsien had accepted prior to his death, an event that of course impacted his research team and collaborators.
"One theme that has gone through all of Roger's work is the desire to peer more closely into the workings of the cell," Adams said. "With all of the fluorescence techniques that he's introduced, he was able to do that in live cells, and make action movies of them in vivid colors. But he always wanted to look closer, and now he's left the beginnings for a method where we can add colors to electron microscopy."
"This is clearly an example of Roger's brilliance at chemistry and how he saw that if we could do this, we would be able to enjoy the advantages of electron microscopy," commented Ellisman, a longtime collaborator who was co-senior author with Tsien on many studies. "The biggest advantage of electron microscopy that we saw is that you have weak contrasts by the nature of the way that staining works so color-specific label give context to all of the rich information in the scene of which molecules are operating."
While the researchers say there is room to improve this technique, it is ready to be used in the lab, and requires tools that are often already available.
If you’d like to know more about the work of Roger Tsien, the video above features Tsien giving a lecture on his work on fluorescent proteins.
Sources:
AAAS/Eurekalert! via
Cell Press News,
Cell Chemical Biology