Finding a Way to Target the 'Undruggable'
Researchers may have made a breakthrough in the fight against a variety of diseases that are difficult to treat. They called their approach a 'kiss of death' that can destroy proteins that have been considered 'undruggable'. The work, performed by scientists at the Unversity of Dundee, has been reported in Nature Chemical Biology. The targeted proteins, which include Ras and Myc, have been linked to cancer and Huntingdon's disease (check out the video below for a discussion about the link between c-Myc signaling and cancer). Led by Professor Alessio Ciulli in the School of Life Sciences at Dundee, investigators have utilized small molecules in a special way; deleterious proteins get bound to neutralizing agents, which initiates a destructive mechanism which eliminates the proteins altogether.
"We know of many proteins which are active in causing diseases, but which we have been unable to block from going 'rogue' or to stop them when they do," explained Professor Ciulli. "The major problem is that we have been unable to find the small molecules which can successfully bind to these proteins and at the same time hamper their function. It is a highly complex area - these proteins can often fool regulators within the cell and be extremely difficult to pin down with inhibitors.
"Research in our lab in the past few years has contributed towards establishing a different approach, one that has been theorized for many years but which is only now fully realized by this latest work. Instead of using the small molecule to try and disable the bad protein, we have developed a way of modifying it so that it can be used to attract the neutralizing proteins, which then bind to their bad neighbor and act against it, starting a cascade process of degradation," continued Ciulli.
"Crucially, we have also found that it is not enough for this neutralizing protein to sit close to the bad protein, it has to make direct contact with it, to 'kiss' it. And not just a little peck, but a real 'Gone With The Wind' embrace. We call this a 'kiss of death,' as it is the key to ensure the degradation of the bad protein," he said.
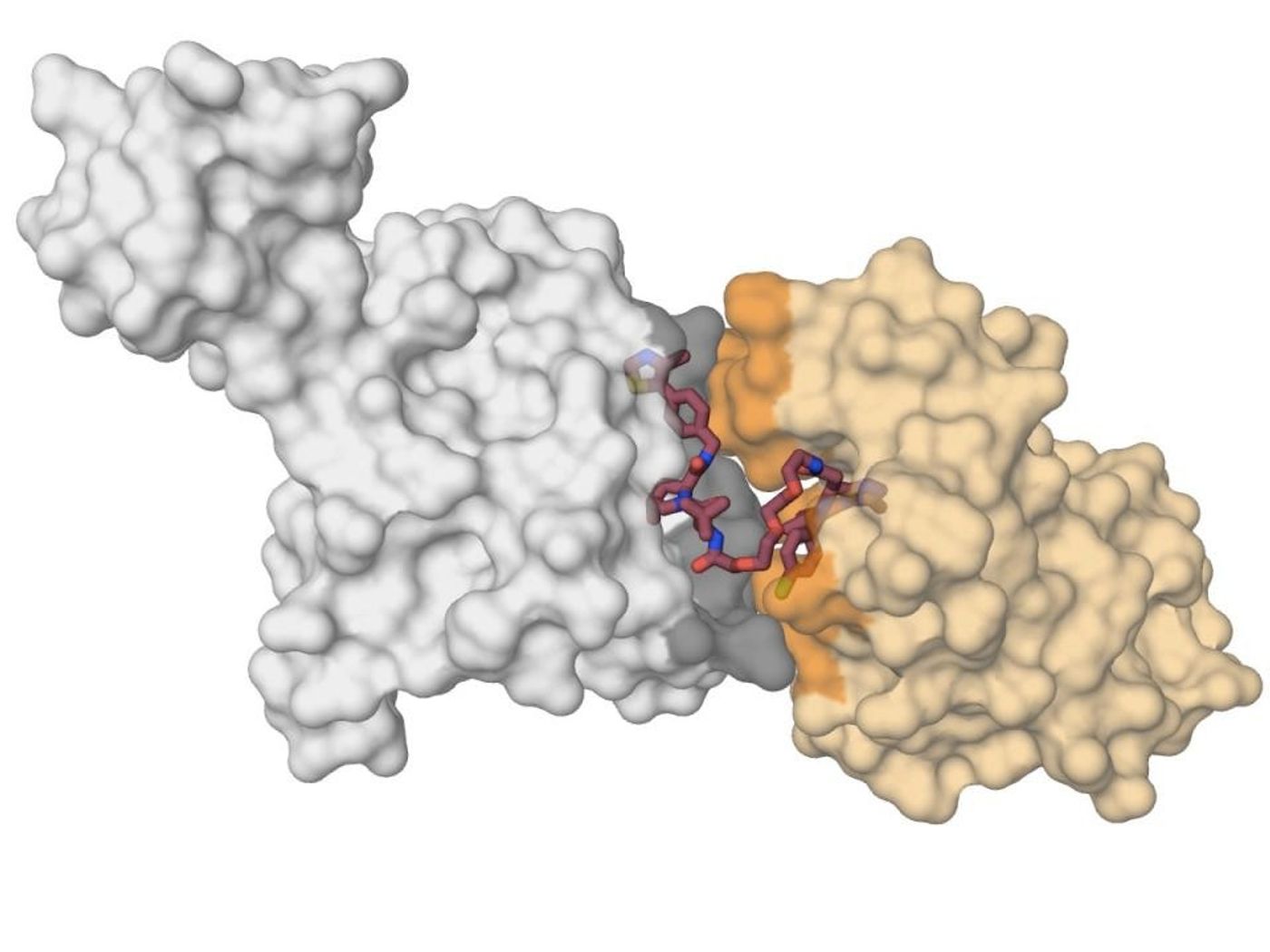
The scientists zeroed in on the proteolysis-targeting chimeric molecule (PROTAC), which can bind a target molecule and the cell's ubiquitin machinery at the same time. The researchers engineered the first X-ray crystal structure of that interaction, showing that PROTACs can be used to bring the target proteins together.
"This discovery provides the first ever insights into how PROTACs work and how we can target proteins for degradation in a highly selective manner," said Professor Ciulli. "This presents a paradigm shift in how we can ensure selective chemical intervention against proteins which we know are factors in causing disease but which until now have been impossible to successfully target. It points towards the possibility of drugging the 'undruggable'."
The investigators paired a candidate cancer drug target with a degrading molecule, specifically the BET bromodomain proteins, BRD4 and a specific BRD4 'degrader,' MZ1. MZ1 folds into itself, drawing the two proteins together. When the target proteins fold in for their 'kiss' the resulting interaction causes degradation of the target. Confirming the specificity of the effect, similar proteins BRD2 and BRD3 don't affect the neutralizing protein. The scientists used that data to create new degraders that spare those proteins from degradation, revealing how such molecules could be designed to minimize non-specific effects.
Professor Ciulli commented that this research is already drawing investment interest from from the pharmaceutical industry. "We now understand better how to turn inhibitors into degraders. The road to turning degraders into drugs will be long and winding and we cannot get there on our own. It is exciting to see signs of serious commitment from the pharmaceutical industry, which adds to optimism that we will be able to get to a point where we can drug the `undruggable'."
The Ciulli lab is highlighted in the video.
Sources: AAAS/Eurekalert! via University of Dundee, Nature Chemical Biology