FDA Warns About Contaminated Supplements
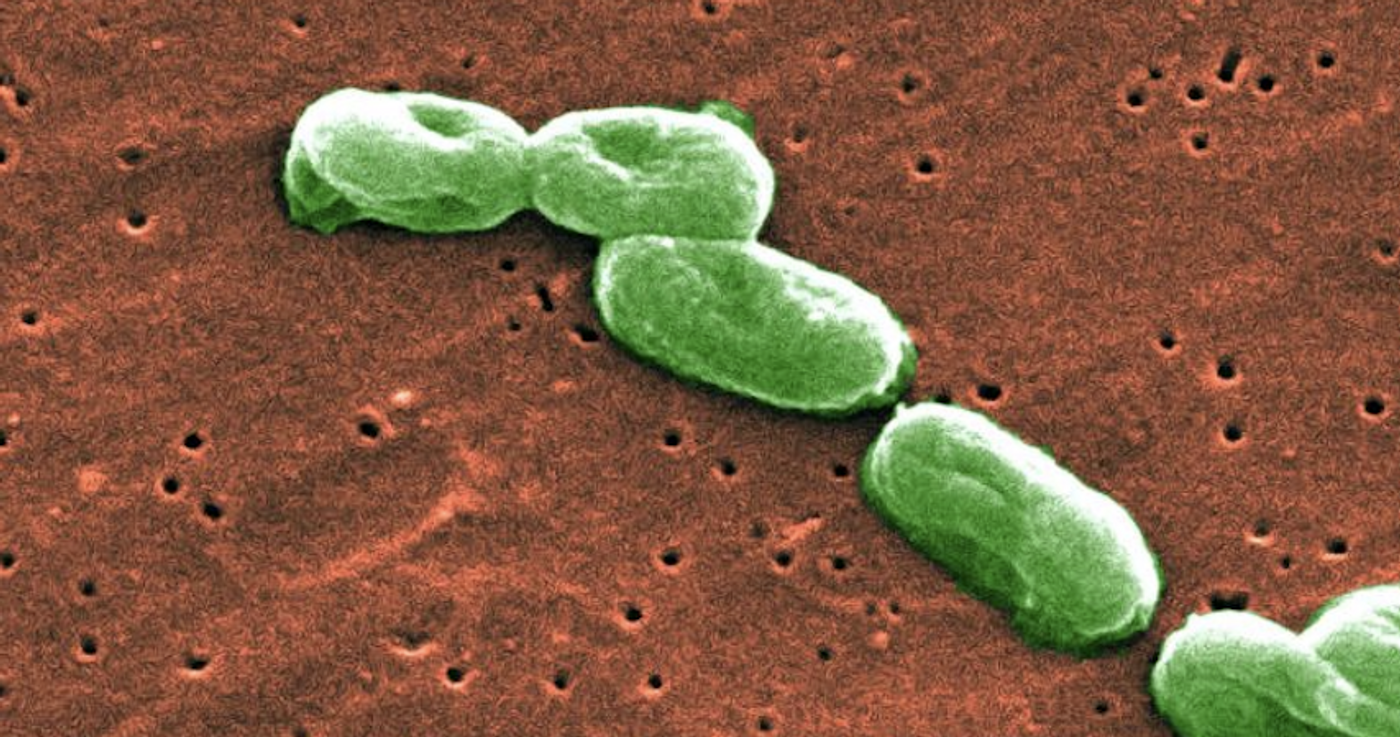
Dietary supplements are popular in the United States. The NIH has estimated that a third of the US population uses some form of supplement. The FDA is now warning consumers about dietary supplements and drugs made by “PharmaTech LLC of Davie, Florida, and labeled by Rugby Laboratories, Major Pharmaceuticals, and Leader Brands,” according to a press release. These drugs could potentially be contaminated with a bacterium, Burkholderia cepacia.
The products included in the FDA’s warning, which is outlined in the video above, include liquid docusate sodium drugs, which are stool softeners. Other products in the warning are dietary supplements such as liquid vitamin D drops and liquid multivitamins for infants and children.
"B. cepacia poses a serious threat to vulnerable patients, including infants and young children who still have developing immune systems," commented FDA Commissioner Dr. Scott Gottlieb. "These products were distributed nationwide to retailers, health care facilities, pharmacies and sold online -- making it important that parents, patients and health care providers be made aware of the potential risk and immediately stop using these products."
Infections with the B. cepacia bacterium can cause a range of symptoms of respiratory infection, or no illness at all. It is contagious though, spreading through direct contact. It also can be resistant to antibiotics. As with many infectious bacteria, it uses the biggest danger to people with compromised immune systems, hospital patients and those that are critically ill.
It's not the first time this manufacturer has caused an FDA warning. The FDA warned both patients and healthcare providers in 2016 to avoid liquid docusate drug products that were made in the PharmaTech facility in Davie, Florida. In that case, there had been a multi-state outbreak of the B. cepacia bacterial infection, and the Centers for Disease Control (CDC) investigated that facility and others.
While manufacturers of supplements do not have to obtain approval before they are released to the market, the Food and Drug Administration regulates these drugs, but under a specific set of rules. These products are considered safe until an adverse event shows otherwise, such as in the 2016 B. cepacia infections. During that event, there were 60 cases in eight states, the drugs were voluntarily recalled, and the outbreak was over by October of that year. The CDC identified PharmaTech as the source of that outbreak.