The Structure of a Bacterial Protein Supercomplex is Revealed
Many different bacteria carry a supercomplex that acts as a kind of battery and helps generate energy. Researchers from the University of Illinois isolated the protein, which sits in the membrane of bacteria and creates a voltage that powers the manufacture of ATP. The ATP molecule is critical to essential processes in many kinds of cells. The scientists are hopeful that this new data, which has been reported in Nature, will aid in the understanding of other membrane protein supercomplexes.
"With billions of years of evolutionary experience, bacteria are adept at surviving in changing environments," said Robert Gennis, a University of Illinois professor emeritus of biochemistry. "Most have the ability to modify, replace or combine molecular tools to suit the new demands - sometimes within a single cell's lifetime," Gennis said. He was co-leader of the work with biochemistry professor Emad Tajkhorshid.
A bacterium takes electrons from food molecules that are high in energy, and uses enzymes to move them to oxygen, in a manner similar to cells of plants or animals, said Gennis.
The random collision of enzymes is what often initiates the movement of electrons. In some cases, enzymes get grouped together by nature, to eliminate the element of chance. Such enzyme supercomplexes can generate voltage, said Gennis, but a particular transfer sequence has to be followed for it to work. That process is a kind of electron transport, a common process used by cells.
"It makes sense that they will function as a single unit to make sure the electron transport is rapid and the electrons end up where they belong," he noted. "Supercomplexes are probably important in all electron transport chains, but in most cases, attempts to isolate them fail because they fall apart. We were lucky to be studying an organism called a Flavobacterium, in which the supercomplex is stable."
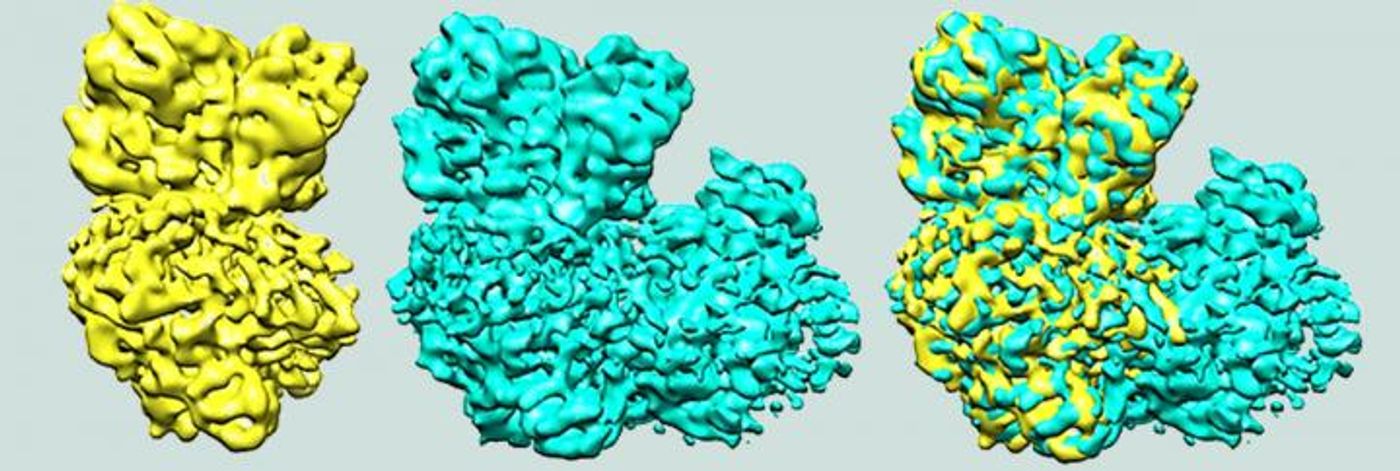
Instead of using detergents to disrupt the membrane (which is a typical method), the investigators removed the protein they were interested in, alternative complex III (ACIII) by using an industrial polymer. That polymer, which is similar to one used to make plastic, enabled the scientists to extract the complex in one swift step.
Cryo-electron microscopy was utilized for this work with the assistance of research groups at the University of Toronto and the New York Structural Biology Center.
"Evolution has resulted in a very efficient nano-machine that is also beautiful to look at. Seeing how this works gives one a great appreciation of nature and is one of the joys of doing science," Gennis said.
Cryo-electron microscopy is described in the video above, from UCSF.
Sources: AAAS/Eurekalert! Via University of Illinois at Urbana-Champagne, Nature