Zapping the nerves to promote nerve regeneration
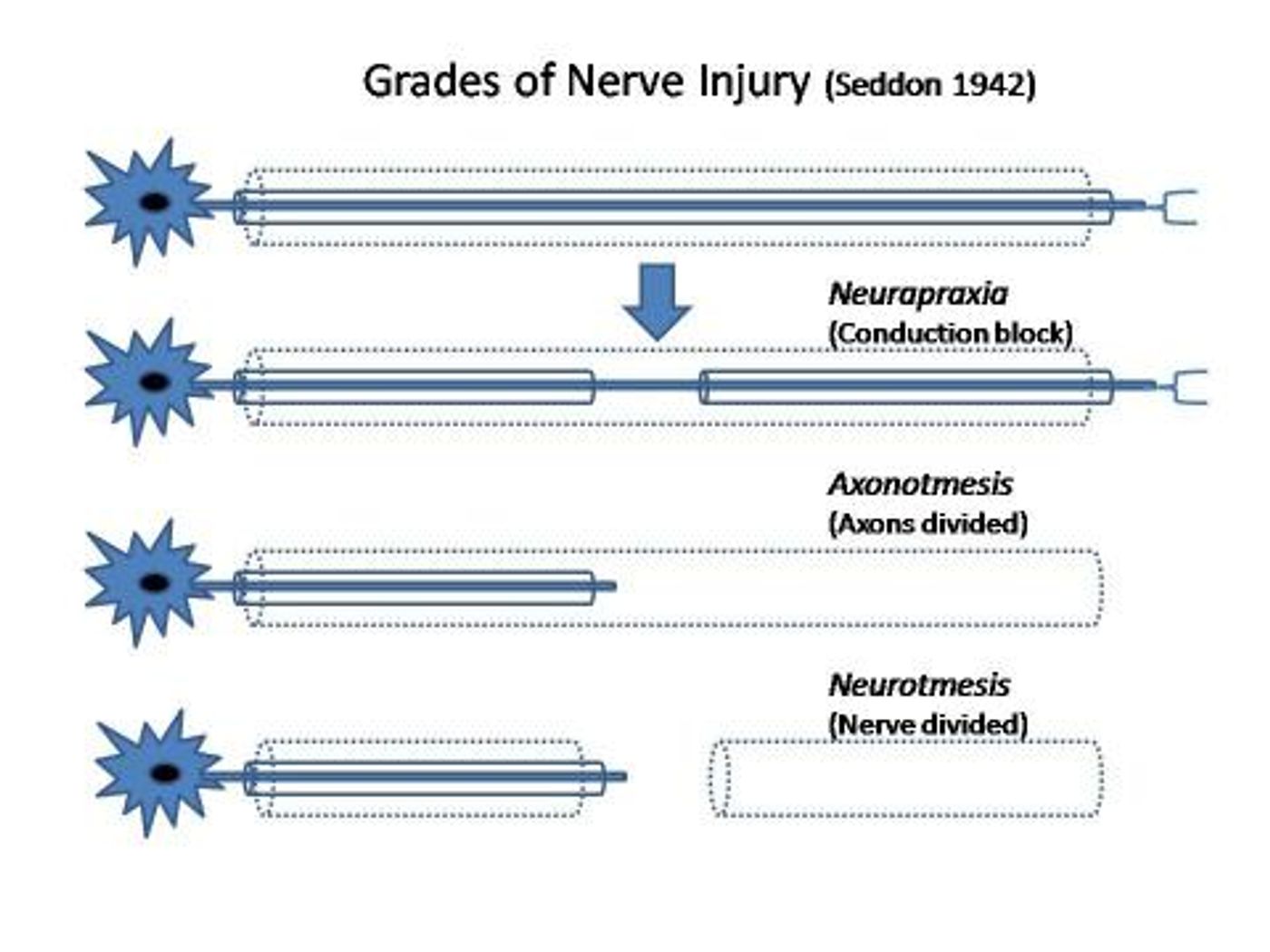
Nerve injury and limb loss are highly prevalent and can have a significant debilitating effect on a one’s quality of life. Unlike other species like gecko’s and salamanders, that can regenerate and replace chunks of their body, we, humans do not possess their abilities. Human peripheral nerves have inherent ability to regrow when injured but is limited. Research on understanding and finding out new ways to improving the regeneration is on the rise. The image below shows the classification of nerve injuries.
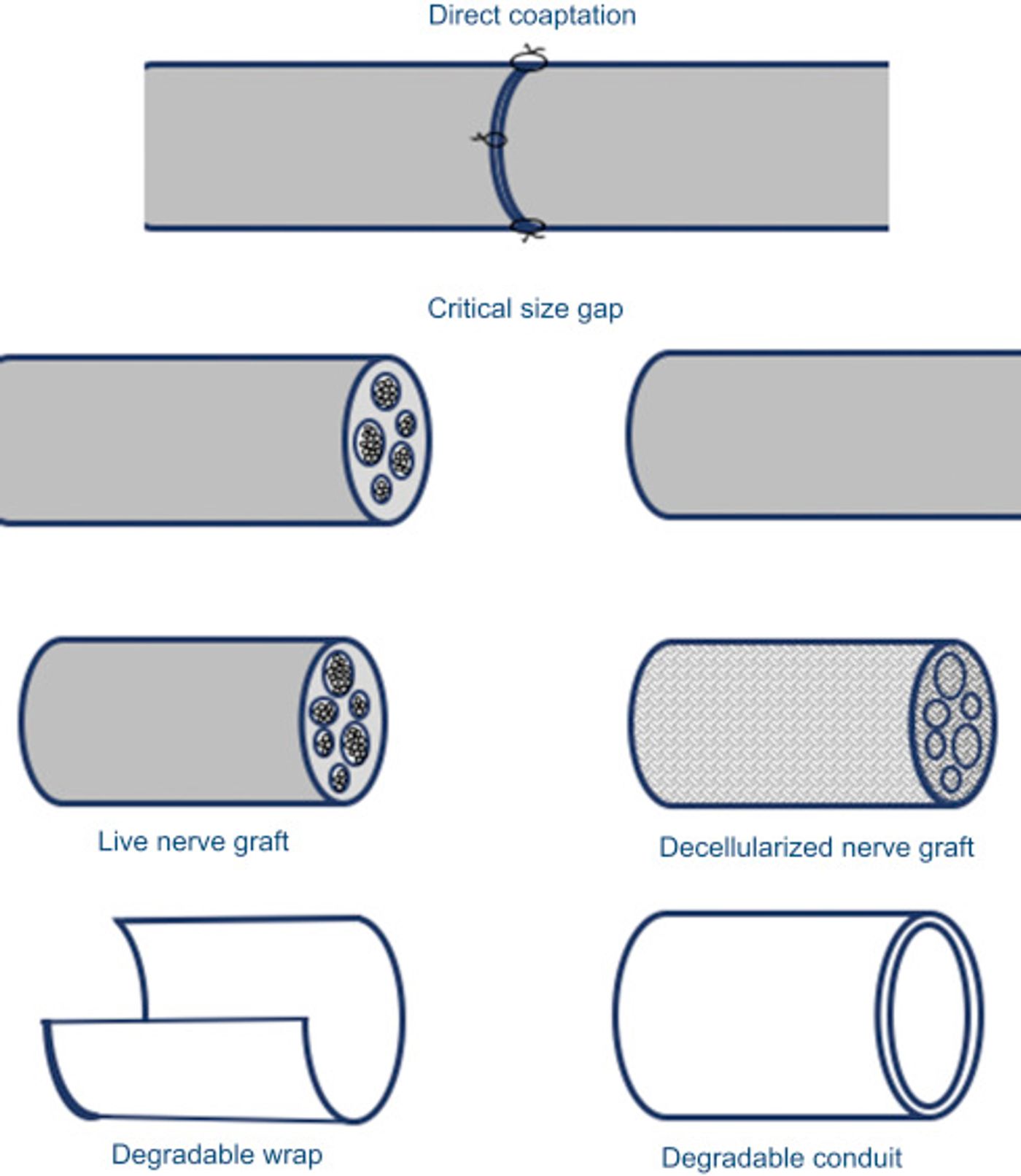
Complete nerve transaction injuries with or without a loss of a chunk of the nerve are the most harder to address. Surgical approaches like end-to-end suturing or using donor nerves to reattach the nerves are the most preferred approaches. However, lack of full functional recovery and donor mismatch/morbidity lead to research on alternative methods. Particularly Nerve transection injuries with a gap of over 3cm, critical nerve gap, between the two ends are much harder to treat. Empty biocompatible conduits are the first choice when a nerve gap needs to bridge. However, misdirection of the growing axons to non-intended targets results in abnormal and / or loss of motor and sensory functions and this further results in a potentially non-reversible cortical plasticity. Alternative strategies to build improved conduits have been explored by many, but the success is still marginal for bridging these critical nerve defects. The image below shows the currently preferred strategies for nerve injuries.
Tessa Gordon, currently working as a senior scientist at Hospital for Sick Children in Canada is one of the pioneers in investigating alternative nerve regeneration strategies based on electrical stimulation. Her early work in 1990’s identified a narrow window upon the onset of injury if intervened, could promote successful regeneration of axons in the peripheral nervous system.
Neurotrophic factors, growth-associated proteins are released post-injury to promote regeneration but these decline overtime and this stunts the axonal growth. Providing exogenous neurotrophic factors that support and encourage regeneration has proved relatively successful over the conventional techniques. However, given the short lifetimes of these proteins, modes of sustained delivery required for successful regeneration and re-innervation of distal targets is still challenging. Furthermore investigating the exact combination and amount of the various trophic factors further complicate their clinical use.
Electrical stimulation (ES) has been used to prevent muscle atrophy post-nerve injury and also promote axonal sprouting in the 1980’s and early 1990’s. Comprehensive evaluation of electrical stimulation induced beneficial effects on nerve regeneration was first described by Gordon’s team in 2000. Brief ES to proximal nerve stumps enhanced axonal growth and prevented de-innervated muscle atrophy ES accelerated nerve regeneration, and these findings were attributed to an increase in the neurotrophin brain-derived neurotrophic factor (BDNF). These findings led to a slew of papers published investigating the multitude of aspects associated with ES therapy for nerve regeneration and a potential clinically translatable therapeutic approach. ES of proximal stump immediate a crush type injury promoted regeneration and successful clinical recovery. To that end, ES of proximal nerve was used to treat patients undergoing carpal tunnel release surgery. A 1-hour low-frequency stimulation (ES) to the proximal nerves enhanced the median nerve outgrowth and their re-innervation of thenar muscles within 6-8 months. In contrast, no re-innervation was seen without the ES.
If one-hour stimulation could do this, then continuous stimulation could promote axonal growth particularly in nerve gaps over 3cm defects. However, challenges associated with wired stimulation devices and the need for multiple surgeries have discouraged successful translation. Now a ray of hope. A bioabsorbable, fully implantable wireless stimulation device that delivers electrical impulses to nerve injuries and promotes regeneration.
“This tape/disc will self-destruct in five/ten seconds,” a recurring quote in mission impossible movies seems to have guided the researchers at Washington University School of Medicine in St. Louis and Northwestern University. The manuscript was published in Nature Medicine with Jahyun Koo from Northwestern University as the lead author.
"We know that electrical stimulation during surgery helps, but once the surgery is over, the window for intervening is closed," said co-senior author Wilson "Zack" Ray, MD, an associate professor of neurosurgery, of biomedical engineering, and orthopedic surgery at Washington University. "With this device, we've shown that electrical stimulation given on a scheduled basis can further enhance nerve recovery."
The device was made from biodegradable materials, and it wraps around the nerve and delivers electrical impulses. A wireless transmitter outside the body powers the device. "These platforms represent the first examples of a 'bioelectronic medicine' -- engineered systems that provide active, therapeutic function in a programmable, dosed format and then naturally disappear into the body, without a trace," Rogers said. "In the case reported here, we built bioresorbable electronic devices that support unique function relevant to recovery from damage to a peripheral nerve, via electrical stimulation at select time points during the healing process."
"Before we did this study, we weren't sure that longer stimulation would make a difference, and now that we know it does we can start trying to find the ideal time frame to maximize recovery," Ray said. "Had we delivered electrical stimulation for 12 days instead of six, would there have been a more therapeutic benefit? Maybe. We're looking into that now."
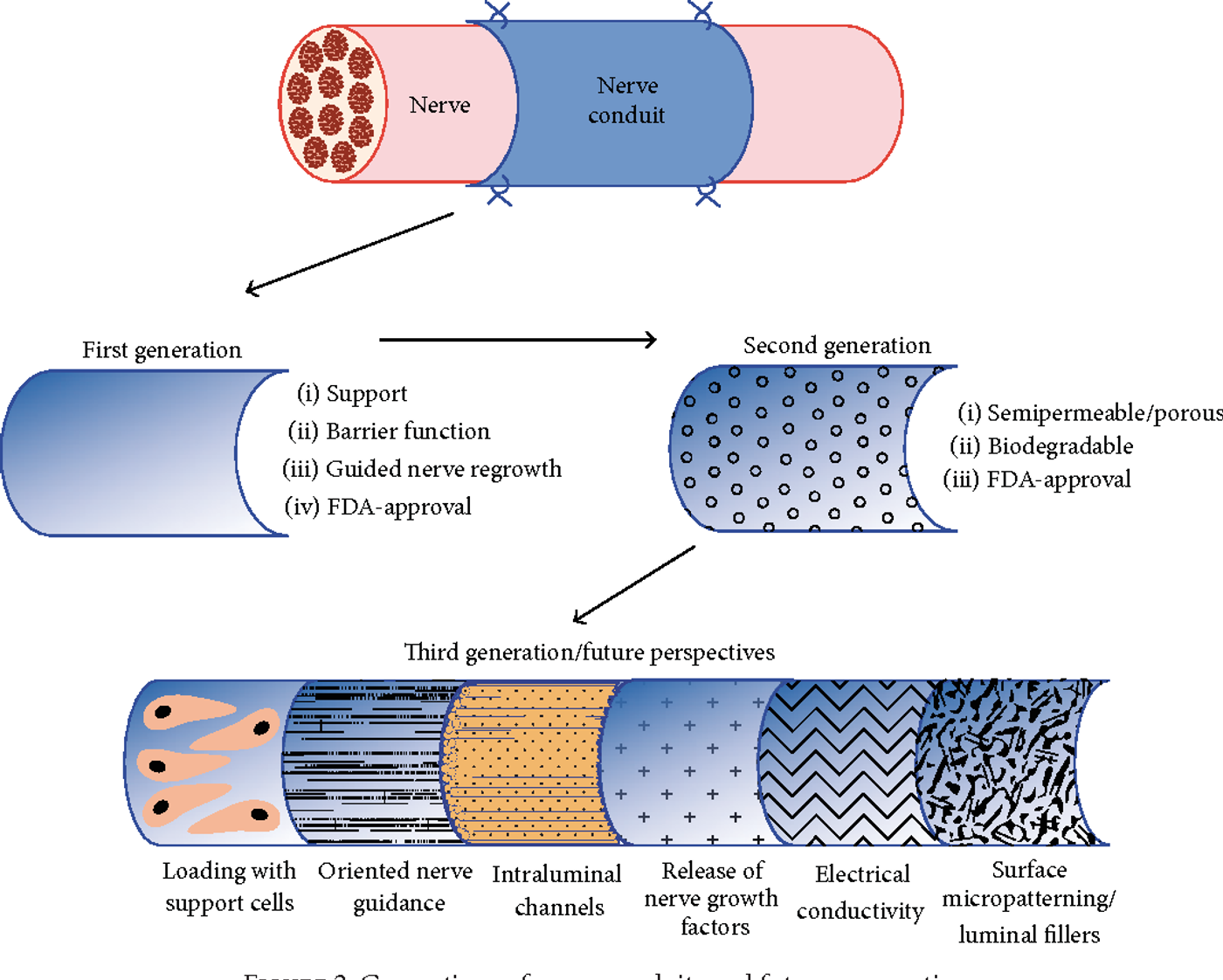
Still, there is a long way to go before humans can mimic gecko’s and rebuild their limbs but on the right track. The following image shows potential strategies to bridge the long nerve gap defect repair.
Sources: Pubmed, Washington University School of Medicine, Nature Medicine, GSK