Gut: a second brain and novel therapeutic target
Gut Microbiome (GM), are considered critical to the development and maturation of the nervous and immune system in humans. Each of our GM has a distinct profile while their distribution along the intestine walls is similar among healthy individuals. The two most abundant bacterial phylotypes present are Firmicutes and Bacteroides and account for at least three-fourths of the entire GM.
David Scheiderer, MD, director of education at Integrative Psychiatry said that’s “All disease begins in the gut.”
An emerging field of research indicates the role of GM in wide range disease pathophysiologies. With time, more and more disease conditions are being added to this. Some of the disease conditions include hepatic encephalopathy, obesity, depression, mood disorders, diabetes, gastrointestinal disorders, and autism among others. Understanding how GM plays a role in pathophysiology could provide novel avenues to diagnose and treat diseases.
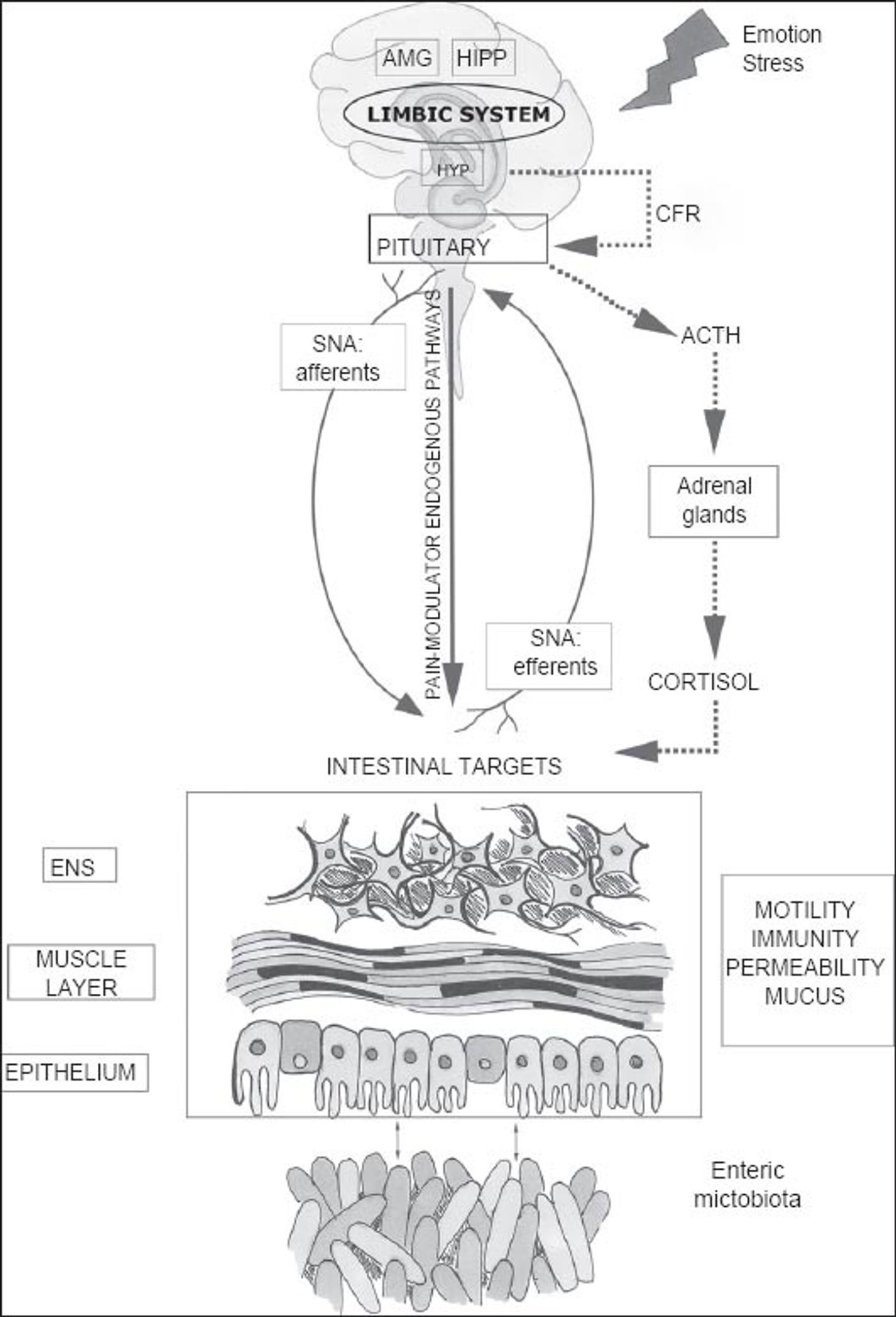
Recently, exploration of pathways by which GM communicates with other physiological systems, and in particular their role in disease pathophysiology, has gained a lot of interest. Gut-Brain axis (GBA), a term coined to elucidate the complex neuro-immuno-endocrine pathways that gut communicates with the nervous system. The image below (adapted from Carabotti et al. 2015), outlines this bi-directional pathway between the central, autonomic, enteric nervous systems and the hypothalamic pituitary adrenal (HPA) axis.
Apart from the hormonal signals to convey messages back and forth from the brain to the intestinal walls, neural signals also contribute to the bidirectional communication. Afferent signals from the intestinal walls through sympathetic and parasympathetic responses are transmitted to enteric, spinal and vagal pathways to brain and efferent from brain to the walls. The stress efferent signals are through the HPA axis.
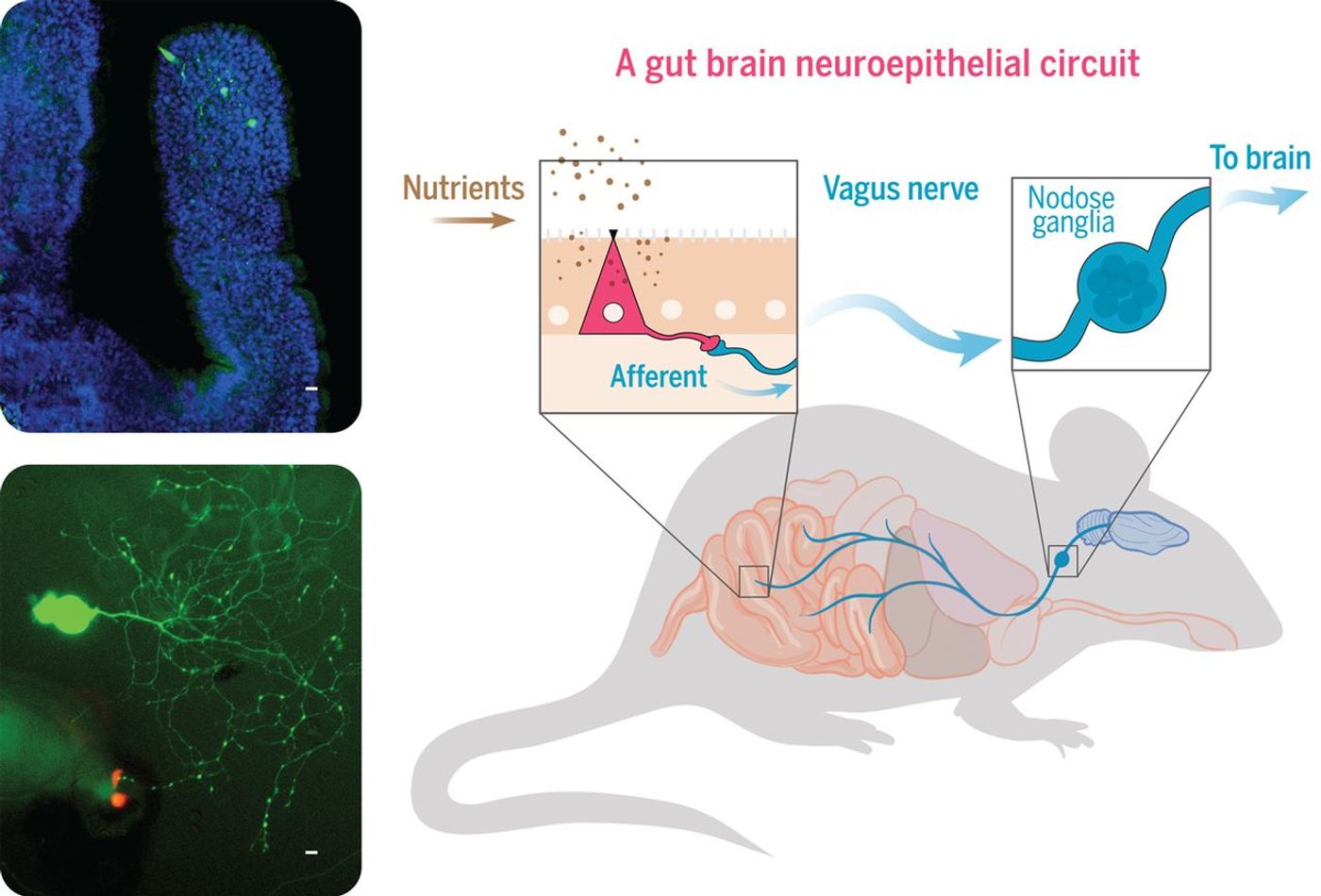
The exact neural circuitry and the transmitters mediating the gut-brain sensory transduction has not been explored in detail. The enteroendocrine cells sense the chemo-attractants and transmit signals to the brain via endocrine hormones like cholecystokinin. However, the neurotransmitter based signal transduction is slow and would not account for the very fast neural responses seen in the vagus nerve when neuroendocrine epithelial cells are stimulated. Dr. Diego V. Bohorquez from Duke University School of Medicine, the senior author on the publication in Science this year, puts vagus nerve higher up on the GBA map. Bohorquez’s team identified endocrine cells that directly synapse with the vagus nerve and termed them neuropod cell. These cells form a neuroepithelial circuit and convey sensory stimulus information from sugars in milliseconds to the vagus nerve. Single cell high throughput RT-PCR assays, western blots enabled this discovery.
The video below elucidates the entire pathway in details.
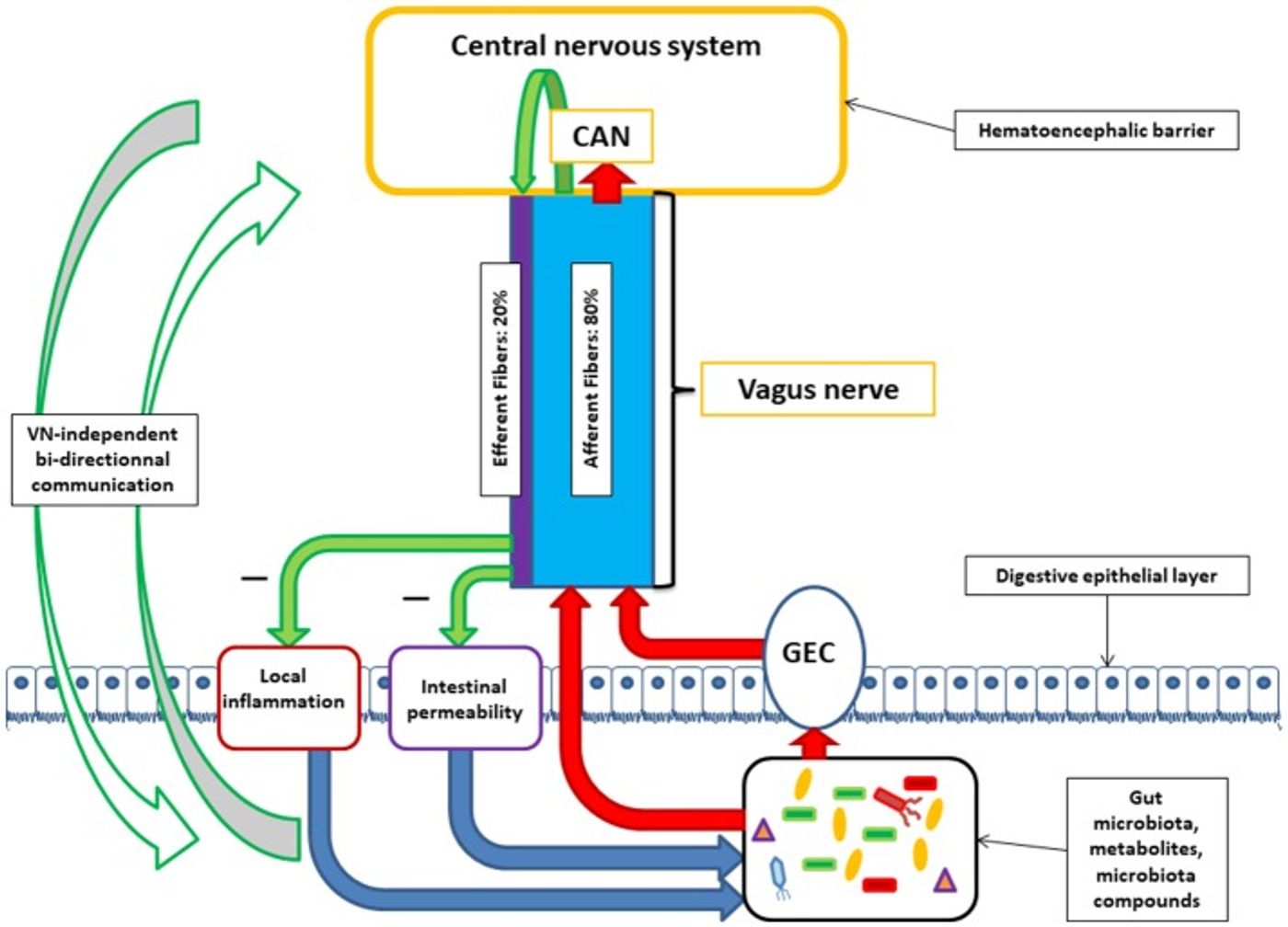
Neuromodulation, a non-pharmaceutical therapeutic strategy that has gained a lot of momentum has been successful in the treatment of arthritis, epilepsy, depression, among others. One unmissable target for successful therapy is the vagus nerve. A recent work published by Bonaz et al., in 2018 Frontiers in Neuroscience explores the possibilities of modulating the GBA via vagus nerve stimulation. Previously described vagus nerve’s role in a cholinergic anti-inflammatory pathway by which inflammatory and intestinal permeability were reduced could potentially involve GM modulation.