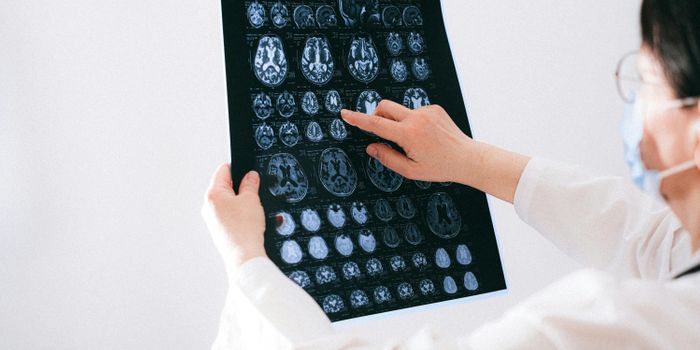
Brain injuries and neurodegenerative diseases are devastating to those who suffer them and incredibly complex for the doctors and health professionals who treat them. There is much that remains unknown about how the brain heals and how to slow the progression of disease. A project being undertaken at Rice University is trying to advance the science of how neurons in the brain form networks and from that data they hope to gain new insights on how the brain can heal itself.
The project at Rice is funded by the
National Science Foundation and is a combination of experiments and number crunching analyses to figure out how the brain organizes the millions of signals and input it receives. The objective is that once it is understood how the brain handles the traffic, develops neuronal networks and processes input these events can then be manipulated to treat brain injuries, strokes and diseases like Alzheimer’s and Parkinson’s.
Researchers and experts from Rice are involved in the areas of nanotechnology, developemental biology and systems biology. Rice bioengineer Amina Qutub, an authority on protein signaling and hypoxia, is leading the effort and she is joined by electrical engineer Jacob Robinson, whose unique microfluidic devices help characterize the electrical properties of living cells, and developmental biologist Daniel Wagner, who develops zebrafish models of human disease. Through the National Science Foundation and
President Obama’s BRAIN Initiative, just over $13 million in grants have been awarded for research into neuroscience.
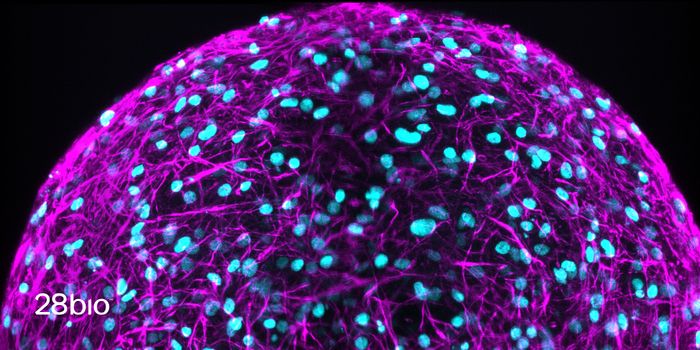
For the first part of the research Qutub and her team will study how neural progenitor cells—the building blocks of neurons—form networks. Electrical activity, chemical signals and spatial patterns must all be looked at through experiments in the lab on human cells to fully understand how the networks within the brain are created.
In a press release from Rice, Qutub said, “We’re focusing on how neural progenitors become functional neural networks. That process is critical for regenerating nerve tissue. There’s still debate over where the progenitors are located and how they’re recruited to areas of injury, but we know they’re present in the brain.”
Once there is enough data on this part of the process, a large computer model will be created to crunch the numbers from the lab trials into a qualitative analysis. Then electrical activity will be measured using a device being developed in the electrical engineering lab at Rice, overseen by study co-lead Jacob Robinson. His method will involve recording electrical activity levels at different points in the development of the progenitor cells to see just when single cells start to form groups or networks. Robinson’s lab will record electrical activity in neural progenitor cells at different points in their development to see how they mature to a functional state.
The third piece of the puzzle is Daniel Wagner’s zebrafish lab where he uses imaging equipment to see into tiny transparent embryos ad record how certain biological processes happen. Wagner has already developed a specific species of zebrafish for the project.
Qutub hopes that what the team learns at Rice can be made available to the global neuroscience community via open-source analysis toolkits. Check out the video below to hear more about this research.