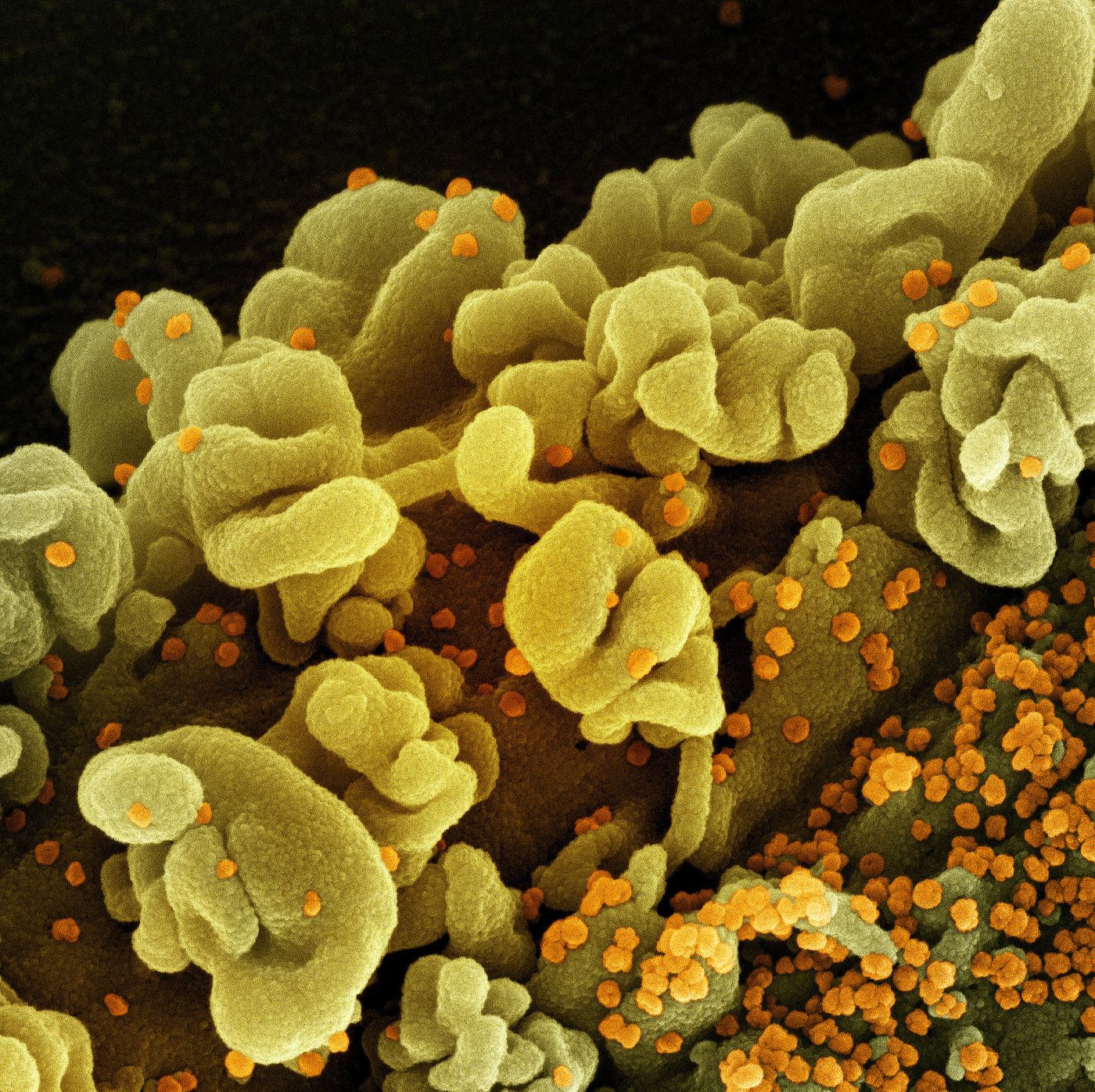
How COVID-19 Can Disrupt the Brain
SARS-CoV-2, the virus that causes COVID-19, does not seem to infect neurons, or if it does, that only happens in rare cases. While the symptoms of a COVID-19 infection can vary widely from one individual to another, severe cases of the infection can have a significant impact on the brain. Scientists have now outlined how the viral infection affected a small group of 40 COVID-19 patients, using CT scans, MRIs, and samples of plasma and cerebrospinal fluid (CSF), and comparing them to a control group. This work has shown that so-called neuro-COVID can disrupt the blood-brain barrier, cause immune disruption in the brain, and can lead to brain structure alterations, seizures, coma, or death in the worst cases. The findings have been reported in Nature Communications.
COVID-19 is known to affect the nervous system; the loss of taste and smell was a common symptom during infections from the original strain of SARS-CoV-2 and early variants, and fatigue has remained a symptom of infection. In this study, the patients had a range of neurological symptoms such as headache and dizziness in the milder cases, acute peripheral neuropathy and myopathy in more serious cases, and stroke, coma, or intracranial hemorrhage in severe cases.
The researchers found that in the group with the most severe neurological symptoms, there had been an excessive immune response. When we get sick, inflammatory processes triggered by the immune system can help us recover, whether it's from a wound or an infection. But that inflammation has to be carefully controlled, or it can become a problem itself. Severely affected patients had impairments in their blood-brain barriers, which the authors suggested was not due to the virus itself, but because of a cytokine storm, in which there is an excessive release of inflammatory signals that are responding to the virus. These patients also carried antibodies that were targeting some of the body's own cells; they had become autoimmune because of that massive immune surge.
"We suspect that these antibodies cross the porous blood-brain barrier into the brain, where they cause damage," suggested corresponding study author Professor Gregor Hutter from the Department of Biomedicine at the University of Basel and University Hospital of Basel.
This work also revealed that the brain's immune cells, called microglia, were causing that excessive immune activation. Patients with the most severe neuro-COVID also had lower volumes in certain parts of the brain such as the olfactory cortex, which controls the sense of smell.
There was also a molecular signature in the blood and CSF that was indicative of an overwhelming immune response in the brain, said Hutter. These biomarkers may one day be used to predict who will develop the most severe cases of COVID-19, neuro-COVID or long COVID, when an infection starts.
The study authors noted that these biomarkers can also reveal targets for COVID-19 treatments. A molecular called MCP-3, for example, is thought to play a central role in the excessive immune response. Hutter said MCP-3 could be inhibited. It should be a priority to prevent an overactive immune response at an early stage of infection, Hutter added.
Sources: University of Basel, Nature Communications