Chemogenetics and optogenetics have been game-changers in neuroscience research. By providing precise control over neural circuits, scientists have been able to use these tools to study behavior and which neurons and circuits are involved in disease in a very in-depth and targeted way. However, as revolutionary as chemogenetics and optogenetics have been, they do have their limitations. Chemogenetics has undesirable kinetics because it takes some time for the injected activator molecule to reach the neurons expressing the receptor and the clearance of the activator molecule can be unpredictable. Optogenetics is much more controlled, but the limitation lies in the fact that light has to reach the neuron being targeted. That makes optogenetics very invasive and limits the ability to study neurons deep in the brain.
Because of how useful but limited these techniques are, a group led by Ali Güler at the University of Virginia have created an ion channel that responds to magnetic fields. The kinetics for this ion channel, obviously dubbed “Magneto,” are fast and controlled, like optogenetics, and using it is noninvasive, like chemogenetics. To build this ion channel, the researchers started with TRPV4, a cation channel that opens in response to stretching of the cell membrane. Exactly how these kinds of mechano-responsive ion channels work is still not known, but it proved to be useful in this construct. To make TRPV4 responsive to magnetic fields, they created a fusion protein with ferritin, an iron storage protein with paramagnetic properties. The idea behind this fusion is that the magnetic field will cause the ferritin to move and pull so that TRPV4 will respond to the pulling by opening and letting Na
+ and Ca
2+ into the neuron. Below is a schematic of how TRPV4 works normally (a) and how the fusion protein Magneto functions (b).
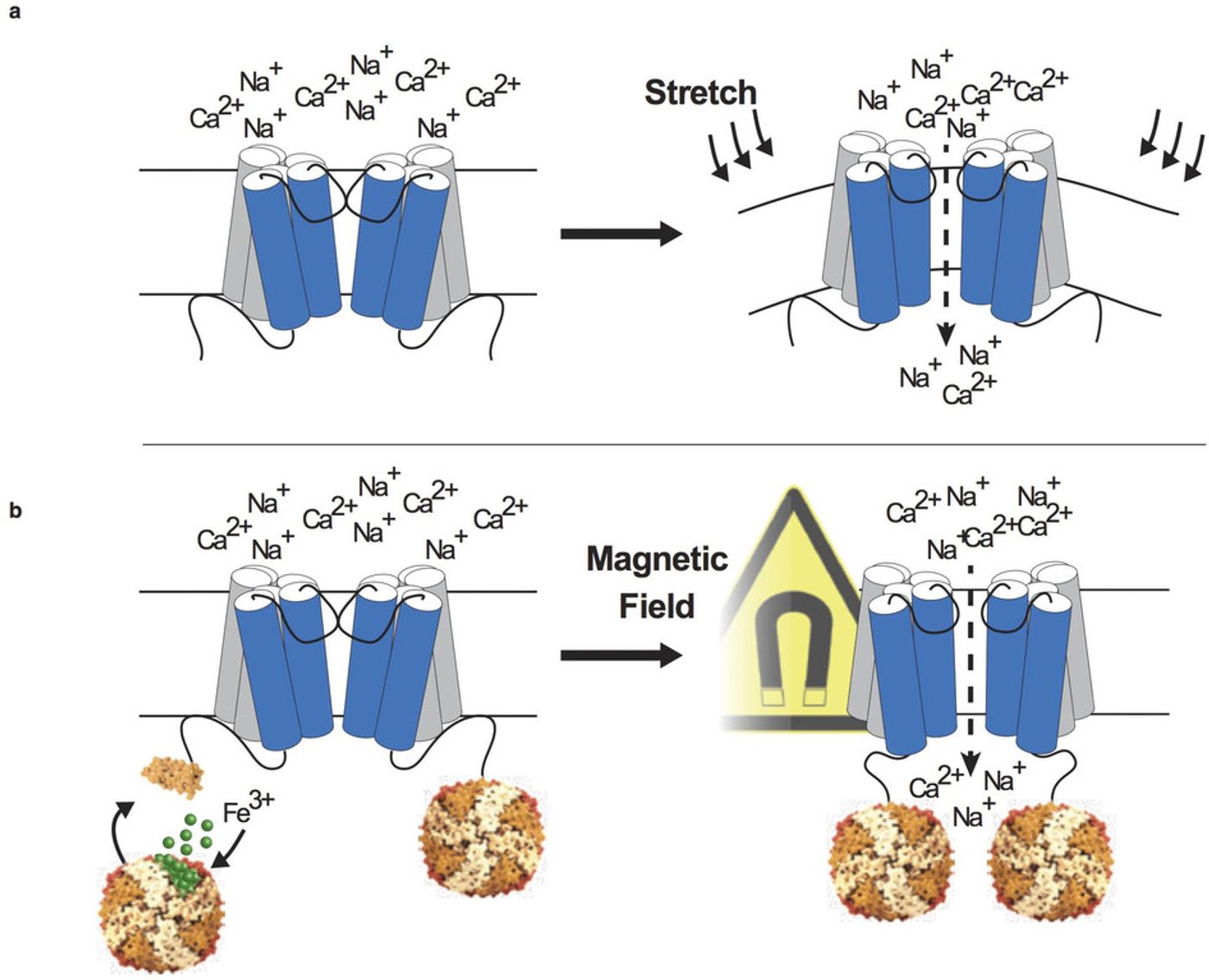
After trial and error and with the addition of a cell membrane-targeting sequence to improve localization and efficacy, the researchers had Magneto. Magneto was tested in multiple models, including mice and zebrafish. Brain slices from mice expressing Magneto in excitatory neurons exhibited rapid neuronal firing in a 50 milliTesla magnetic field and abruptly stopped firing when the magnetic field was turned off. 50 milliTeslas is stronger than a refrigerator magnetic but perfectly safe. For reference, an MRI machine produces a magnetic field between 1 and 3 Teslas. In zebrafish, Magneto was expressed in a specific type of neuron, the Rohon-Beard sensory neurons, which cause the zebrafish larvae to curl up when stimulated. In the presence of a magnetic field, the zebrafish larvae curled up 10 times more often than larvae not in a magnetic field. In another mouse construct, Magneto was expressed in medium spiny neurons in the striatum which are involved in reward behavior. Mice were allowed to explore two chambers, one with a magnetic field and one without. Wild-type mice showed no preference between the two chambers, but the mice expressing Magneto showed a significant preference for the magnetic field chamber. The magnetic field opened Magneto to stimulate the medium spiny neurons which activated a reward circuit that made the mice prefer the magnetic field.
These first experiments with Magneto look very promising, but the group already has ideas for how it can be improved. TRPV4 has ligand binding sites, so one way to improve the specificity of Magneto would be to get rid of these binding sites so that the only thing that could activate Magneto is a magnetic field. Even though Magneto in its current incarnation is a prototype, it has the potential to be a game-changing research tool like optogenetics.
Sources:
AlzForum and
Nature Neuroscience