Glutamate is the main excitatory neurotransmitter and is essential for many important processes in the brain. In accordance with glutamate’s importance in the brain, there are many different types of glutamate receptors, capable of relaying glutamate’s message in a variety of ways. There are metabotropic glutamate receptors designated as mGluR that communicate through G-protein second messenger signal cascades. There are also three separate types of ionotropic glutamate receptors (AMPA, kainate, and NMDA receptors) that are ion channels capable of very rapid signal transduction. NMDA receptors in particular are very important for the induction of long term potentiation, a process that is crucial for learning and memory. Compared to the other two kinds of ionotropic glutamate receptors, NMDA receptors have relatively slow kinetics; they are slow to open and they are slow to close in response to excess synaptic glutamate. Dysfunction of NMDA receptors is involved in stroke, schizophrenia, epilepsy, Alzheimer’s disease, and anti-NMDA receptor encephalitis.
Because of their involvement in so many neurological disorders, it is really important to understand how these receptors work. NMDA receptors are mostly heterotetrameric, meaning they consist of four subunits that are not all the same. The NMDA receptor will always have two NR1 subunits, but the other two subunits will be some combination of NR2(A-D) and NR3 subunits. Until recently, all crystal structures of the NMDA receptor were made by substituting a cysteine for a lysine to create a disulfide bridge and reduce mobility of the receptor. While this substitution made getting a structure easier, it also made it much less physiological, which is why a recent paper published in
Cell is so impressive. First author Shujia Zhu et al. from Rockefeller University used cryo-electron microscopy (cryo-EM) and double electron-electron resonance (DEER) to get a more detailed and physiological structure of an NMDA receptor bound to agonists and antagonists.
What Zhu et al. found was very different from the previously reported structures created with the cysteine mutation. In the K216C (lysine at residue 216 mutated to cysteine) receptor, there was a lot less range of motion in the amino- terminal domains (ATDs) of the NMDA subunits compared to the new structures. When a competitive antagonist (a molecule that binds to where the agonist would bind and prevents the receptor from being activated) is bound to the NMDA receptor, the ligand binding domains of the NR2 subunits “flip” and there is a lot of separation between the ATDs. The researchers were not expecting to see this kind of dramatic conformational change in the receptor.
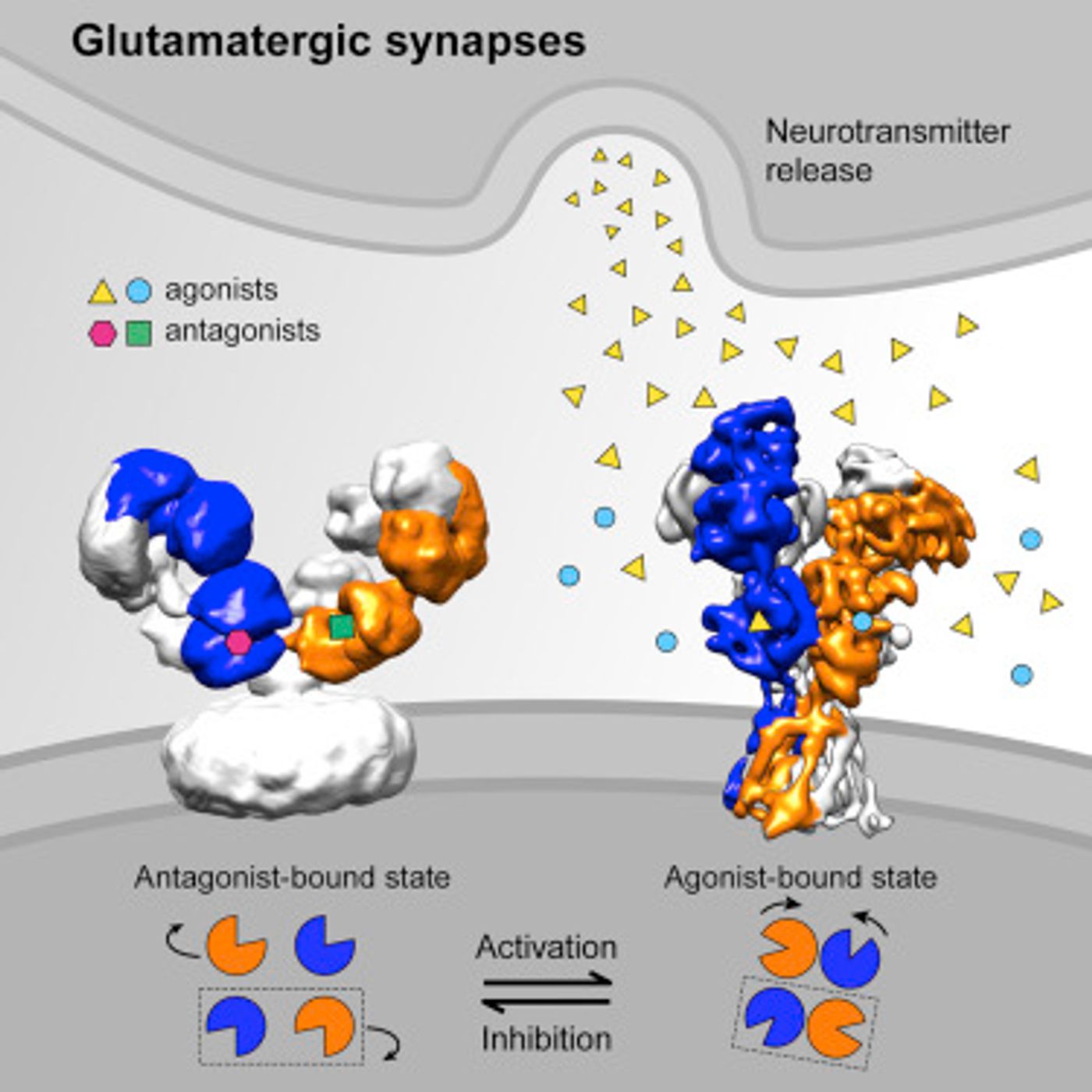
They were also not expecting to see a similar conformation in the desensitized state. Since excess glutamate neurotransmission through ionotropic receptors can very easily cause excitotoxicity, NMDA receptors desensitize in response to excess synaptic glutamate. Basically, even though the agonist (glutamate) is bound, the receptor closes and does not allow any more ions into the neuron. In the NMDA receptor’s desensitized state, there is a lot of spatial separation between the ligand binding domains of the various subunits, similar to the antagonist-bound state. This is a huge difference compared to the AMPA receptor whose desensitized conformation does not disrupt the interface between ligand binding domains.
Elucidating these kinds of differences may not seem like a big deal, but the speed of NMDA receptor desensitization has clinical relevance for epilepsy and Alzheimer’s disease. Understanding how the receptor changes shape and how that affects the time course of desensitization increases our understanding of disease pathology and also gives us more information about how learning and memory are encoded in the brain at the molecular level.









