Binge drinking is a huge problem in the United States, with 1 in 6 adults binge drinking four times a month. Costs due to binge drinking were $191 billion in 2010 in the form of lost productivity, health care, crime, and other expenses. For the most part, those who engage in binge drinking are not alcohol dependent, but repeated binge drinking episodes are part of the transition from moderate, controlled alcohol use to alcohol dependence. Research on how binge drinking affects the brain is important because it could lead to the identification of pharmacological targets that could curb binge drinking and prevent the transition to alcohol dependence.
A group at University of North Carolina Chapel Hill has recently published a paper in
Biological Psychiatry that details a functional circuit involved in modulating binge drinking behavior in mice.
First of all, what qualifies as a binge drinking episode and how would you model that in mice? The National Institute of Alcohol Abuse and Alcoholism defines binge drinking as “a pattern of drinking characterized by consuming enough ethanol to rapidly achieve blood ethanol concentrations (BECs) >80mg/dL (0.08%).” This is ~5 drinks in 2 hours for men and ~4 drinks in 2 hours for women. There is actually a very well-established protocol for modeling binge drinking is rodents called the drinking-in-the-dark (DID) procedure. For 3 days, the water bottles in the mouse cages are switched out for a 20% unsweetened ethanol solution for 2 hours starting 3 hours into the dark cycle (nighttime in the animal facility). On the fourth day, the mice are given the ethanol for longer, between 2-4 hours, and this is the binge test. Following this procedure, mice will often drink enough to have a BEC greater than 100mg/dL which is sufficient to produce significant motor impairment and a tolerance to this effect over repeated binge drinking episodes.
By using the DID model of binge drinking, the researchers were able to identify specific neurons in the bed nucleus of the stria terminalis (BNST, a major output from the amygdala) that project to the ventral tegmental area (VTA) that exhibit dysregulated signaling following binge drinking episodes. Specifically, they determined that corticotropin releasing factor (CRF) signaling modulates binge drinking. CRF, a small peptide, binds to two G-protein coupled receptors (GPCRs), CRF1R and CRF2R. Based on the evidence they gathered, the researchers hypothesized that the CRF-releasing neurons that project from the BNST to the VTA modulate GABAergic signaling in the VTA by binding to presynaptic CRF receptors and that is what controls the binge drinking behavior. Specific pharmacological inhibition of CRF1Rs in the VTA lessened binge drinking behavior, but that effect was dependent on unaltered CRF2R signaling.
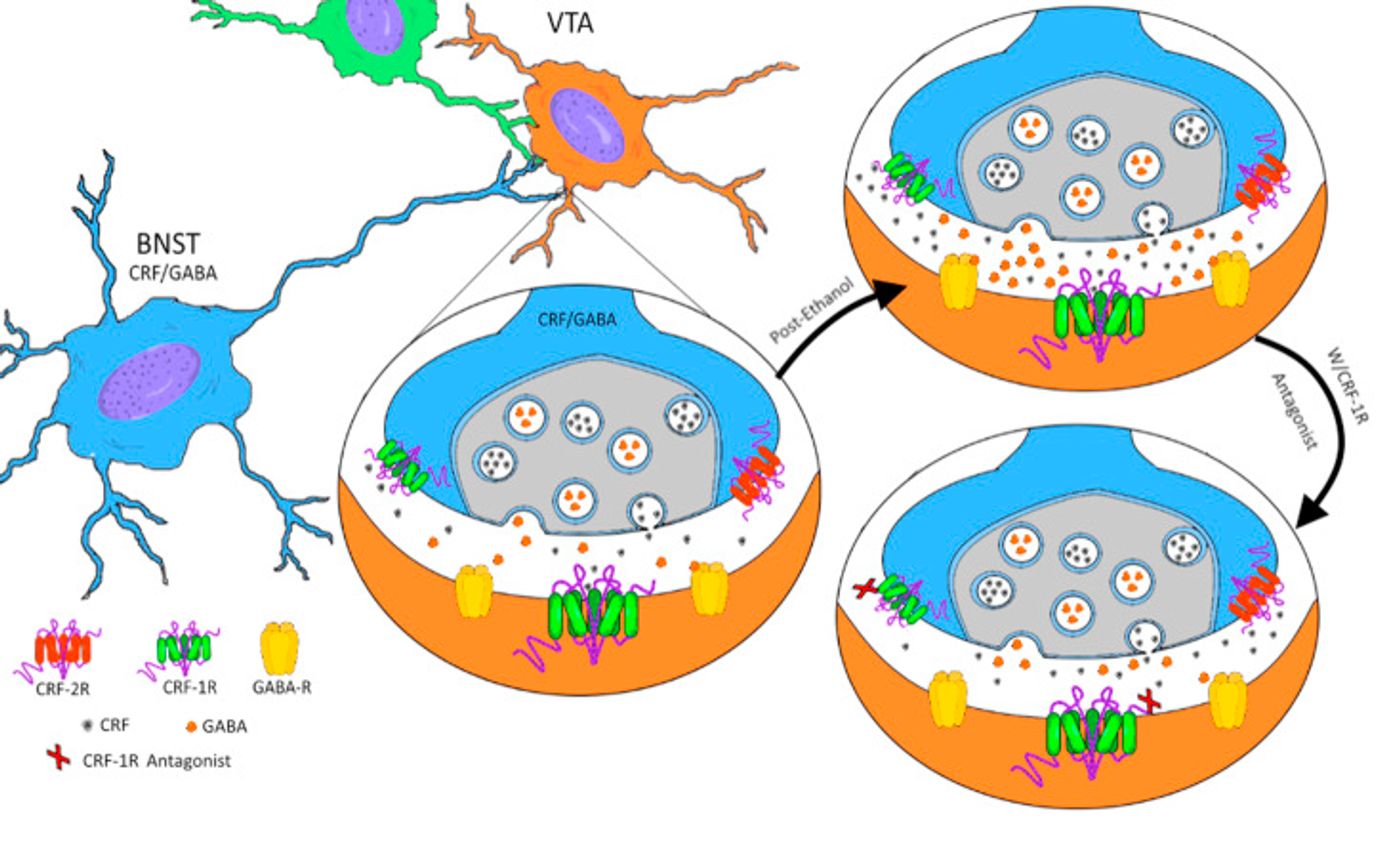
This paper is the first time that a functional circuit from the extended amygdala to the VTA has been described and characterized. Hopefully this circuit will continue to be studied and will one day be used to create pharmacological treatments for binge drinking and to prevent the transition from binge drinking to alcohol dependence.
Sources:
EurekAlert,
Biological Psychiatry,
DID procedure, and
CDC