New Street Drug Alert: Kratom
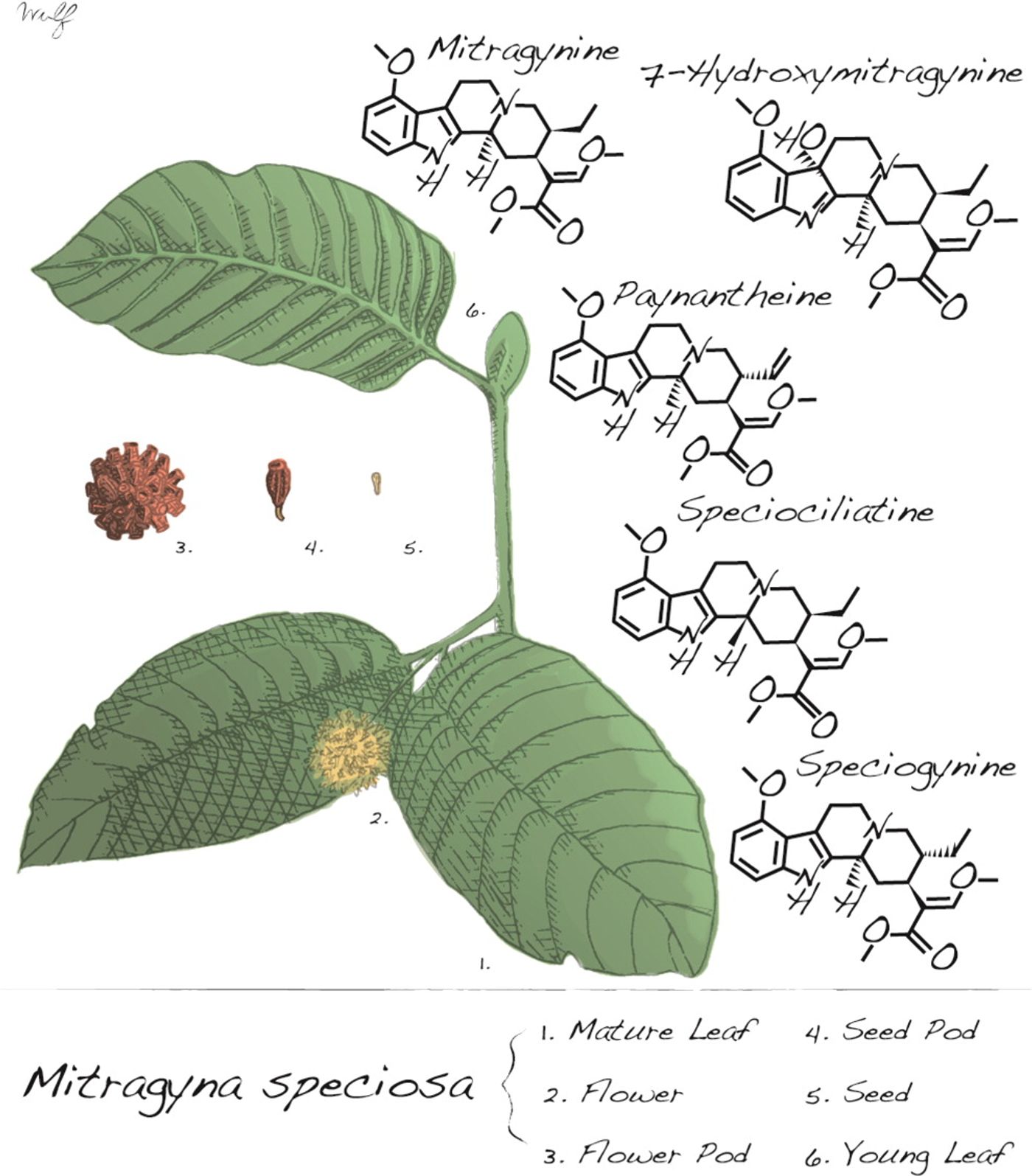
There has been an increasing amount of attention on an interesting, legal drug known as kratom online that purportes to help ease the difficulty of heroin withdrawal and treat chronic pain and depression. Kratom (Mitragynia speciosa korth) is a tropical tree indigenous to Southeast Asia whose leaves are chewed or dried and powdered. Recreational use of kratom is relatively new in the U.S. and Europe, but has been used in cultural ceremonies, as a stimulant for laborers, and to treat heroin addiction for many years in Southeast Asia. Thailand actually started regulating kratom in 1943 as heroin became more expensive and users adopted kratom as a cheaper alternative.

Kratom has a very interesting pharmacological profile because it exhibits both stimulant and opioid-like effects in a dose-dependent manner. A low dose acts as a stimulant while a higher dose acts as an opioid. A recent paper published in the Journal of the American Chemical Society showed kratom binding to human μ opioid receptors (MORs) for the first time in vitro. Opioids such as morphine, heroin, and fentanyl bind to MORs, but kratom is different in that it is less potent and has a biased response.
The kratom leaf contains over 25 different alkaloids, with the concentrations of each alkaloid varying between the different strains of kratom tree. Even with the variation, two alkaloids in particular, mitragynine and 7-hydroxymitragynine (7-HMG), are thought to cause the majority of the psychoactive effects. About 60% of the total alkaloids present in the kratom leaf extract is mitragynine, and 7-HMG accounts for about 2%. Mitragynine by itself only has about 1/100th of the binding potency of morphine at human MORs as assessed in non-neuronal cells overexpressing human MORs, but it is possible that kratom extracts contain enough of the compound to have a psychoactive effect or that the binding potency is higher in a more physiological setting. 7-HMG is 10 times more potent than mitragynine at MORs and interestingly, storage conditions can have an effect on the conversion of mitragynine to its more potent metabolite, 7-HMG. Kratom extract stored in oxidizing conditions with access to sunlight converted about half of the mitragynine to 7-HMG. Sunlight alone was able to convert about 8% of mitragynine to the more potent compound.
Since research on kratom is relatively new, there is no consensus of the addictive and toxic properties of kratom. As a result of this, and because kratom is not sold for human consumption, it is in a similar legal position as synthetic cathinones (bath salts) and synthetic cannabinoids.
From a research perspective, the cocktail of alkaloids found in kratom could hold some incredibly valuable information. The Journal of the American Chemical Society paper showed that the major constituent alkaloids of kratom extract bind to and partially activate human MORs. Most importantly, mitragynine and 7-HMG bound to MORs but did not recruit β-arrestin. β-arrestin recruitment at MORs is responsible for all of the undesirable effects of classic opioids like morphine, including constipation, respiratory depression, and the development of tolerance. Researchers could exploit the biased response of the kratom alkaloid scaffold to create safer painkillers and to potentially create better treatments for opioid addiction. Considering that opioid overdose is the leading cause of accidental death in the U.S., this is imperative.
Sources: J Am Chem Soc, New York Times, Scientific American, and Int J Legal Med