Synapses are the connections between neurons that allow them to communicate with each other. When synapses become compromised and can no longer effectively communicate, they are lost, and brain function suffers. Synaptic density would be a great measure for diagnosis and for tracking neuropathology in certain diseases, but there is currently no way to look at synaptic density in the living brain. Evaluating synaptic density requires brain tissue from autopsy or surgical resection. However, scientists have developed a new radioligand for PET (positron emission tomography) scans that is capable of measuring synaptic density in the living brain.
This research was recently published in Science Translational Medicine and described PET studies with a new radioligand that were conducted in non-human primates, healthy human subjects, and epileptic patients with mesial temporal lobe sclerosis.
Usually when measuring synaptic density in brain tissue, researchers will use an antibody against synaptophysin, a protein that is present in every single presynaptic vesicle. However, there is currently no radioligand that can be used for PET that binds to synaptophysin. The company
UCB does have an antiepileptic drug, levetiracetam, that targets another synaptic vesicle protein, SV2A. A derivative of this drug, [11C]UCB-J, is the radioligand that was tested in these studies and has been shown to specifically bind to SV2A. SV2A, just like synaptophysin, is present in every synapse.
When they tested [11C]UCB-J in non-human primates, they saw a signal in gray matter and none in white matter, which makes sense. Gray matter consists of the axons and dendrites of neurons, i.e. where the synapses are, and white matter is the cell bodies of those neurons. To investigate the quantitative nature of their tracer, the scientists also cross-validated PET signals with post-mortem brain tissue analysis of SV2A protein levels in several brain regions.
Following non-human primate studies, [11C]UCB-J was tested in healthy human subjects and was found to be effective and had favorable kinetics. The real test was to do a PET scan with [11C]UCB-J in epileptic patients with mesial temporal lobe sclerosis. In patients with this condition, there is loss of synapses in the hippocampus in one hemisphere and the atrophy is also visible on MRI. With this new tracer, the scientists were able to visualize the loss in synaptic density in the hippocampus on the affected side compared to the unaffected side, and were also able to correlate the synapse loss with degree of atrophy.
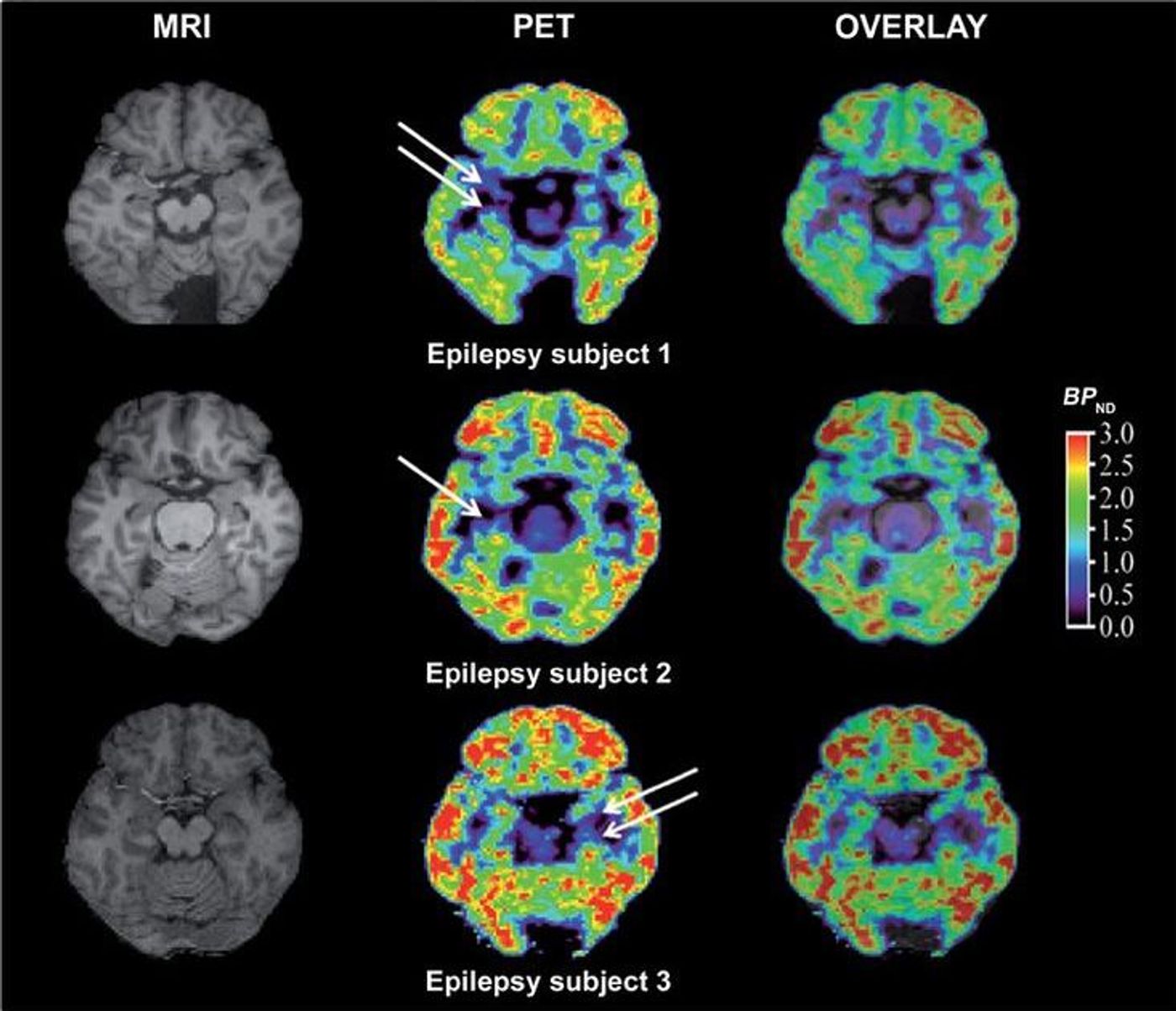
This is a crazy advancement for neuroimaging because synapse loss is present in so many neurological conditions and is also one of the pathological events in Alzheimer’s disease (AD) that closely tracks with cognitive decline. When neurons die, they die, and there is no getting them back. But it might be possible to reverse damage to the synapses which is the step that precedes neuronal loss in AD. Which this kind of imaging, it will be possible to monitor the effect of early interventions in AD on synaptic density, a measure that correlates with cognitive decline more so than other biological markers.
I have barely scratched the surface, and if you are interested, I strongly suggest looking at the original article in
Science Translational Medicine and also this great write-up on
AlzForum.