Genomic Markers Identified for Infant Soft Tissue Tumors
Epidermal Growth Factor Receptor (EGFR) helps maintain cellular growth and division normally but can become mutated, as in the case with some cancers, so that too many receptors allow exponential, uncontrolled cellular growth and division resulting in a tumor. EGFR is a transmembrane kinase glycoprotein; its gene locus is found at 7p11.2 and is commonly associated with multiple different cancers as part of gene rearrangements whereby the EGFR signaling pathway becomes deregulated. EGFR is implicated and/or found in many cancers including lung, breast, head, neck, and colorectal.
B-Raf is another kinase protein associated with processing mitogenic signals between the cell membrane and the nucleus of a cell and its enzymatic activity has been associated with Epidermal Growth Factor and Human Growth Factor ligands. The gene BRAF is located at 7q34 and is considered a proto-oncogene; there is significant literature outlining the present of mutations and rearrangements involving BRAF resulting in a multitude of cancers including melanoma and colorectal cancer.
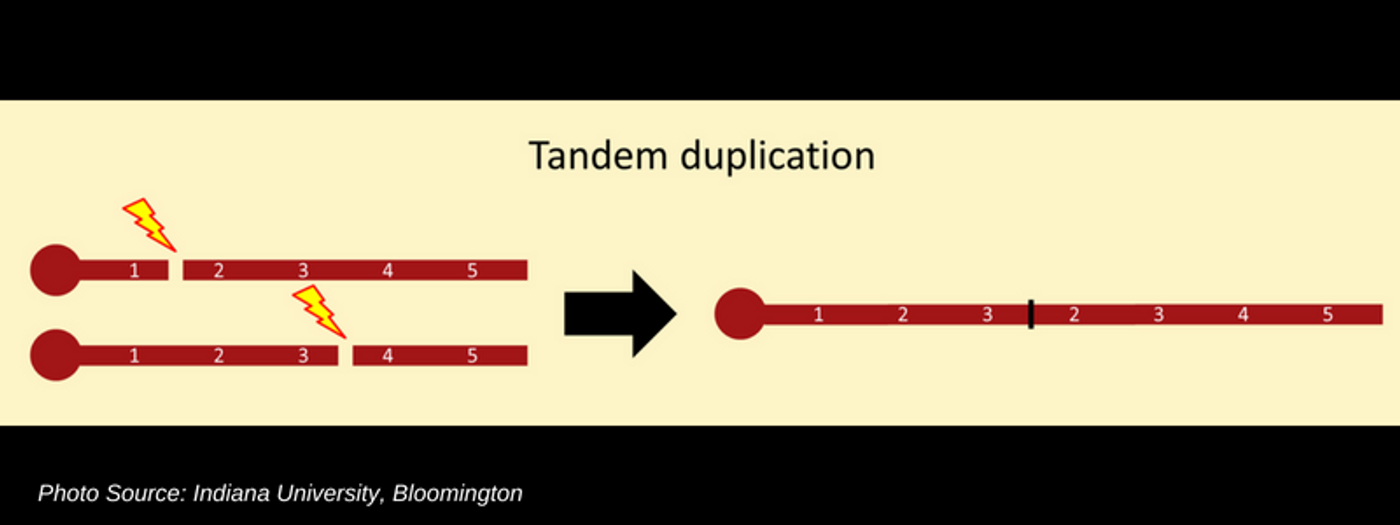
A new article from Nature Communications published on June 18, 2018 now outlines a newly discovered connection between multiple different infant soft tissue tumors and these two specific genes. Childhood tumors, especially those in infancy are often confined to age and developmental stage but little has been known about their etiology and therefore, how to best treat them. Congenital mesoblastic nephroma (CMN) is renal malignancy in children under 6 months old. Infantile fibrosarcoma (IFS) is comprised of mutated spindle fiber cells often in the soft tissues of the extremities or abdominal region in an infant. The researchers examined similar soft tissue tumors for the presence of EGFR or BRAF rearrangement as a diagnostic marker and found that in all CMN specimens evaluated, there was a single in-frame internal tandem duplication (ITD) within the EGFR domain. The EGFR-ITD was not observed in other tumor types like glioma or adenocarcinoma of the lung. Additionally, the group discovered mutations in the BRAF gene in some CMN cases also. A mutated fusion protein as a result of BRAF rearrangement has been observed in some IFS patients but has not been seen in CMN.
Genomic findings can be groundbreaking for the course of cancer treatments used and for the future development of novel treatments; basing clinical and therapeutic decisions on the mutations present, rather than on the phenotypic findings, is changing the practice of oncology. For infants with CMN and EGFR-ITD, already existing treatments are available which target EGFR mutations previously identified in other cancers. This is also true for IFS where a BRAF mutation is the driver.
Sources: Nature Communications, Oncology Reports, Wellcome Sanger Institute, American Society of Clinical Oncology, Expert Opinion on Therapeutic Targets,