When the equilibrium of gene regulation is disrupted, whether by random mutation, ultraviolet light exposure, or another cause, the risk of abnormal cell division and tumor growth drastically increases. A new study from the University of Leicester describes a way to restore equilibrium and reduce incidence of genetic diseases.
MTA1, RBBP4, and HDAC1 are core protein components of a complex called NuRD, which is responsible for regulating the amount of protein made from transcription and translation processes. When these core proteins’ interactions are inhibited or communication is misconstrued, the complex falls out of equilibrium and diseases can sprout and progress.
NuRD regulates gene expression through the control and maintenance of chromatin, the genetic material that forms chromosomes inside eukaryotic cell nuclei (
Nature). Chromatin allows for DNA to be organized into tightly-wound complexes called histones, which then form nucleosomes. This highly-ordered structure is dependent on consistency and accuracy, so when one brick is out of place, the whole system struggles to function properly.
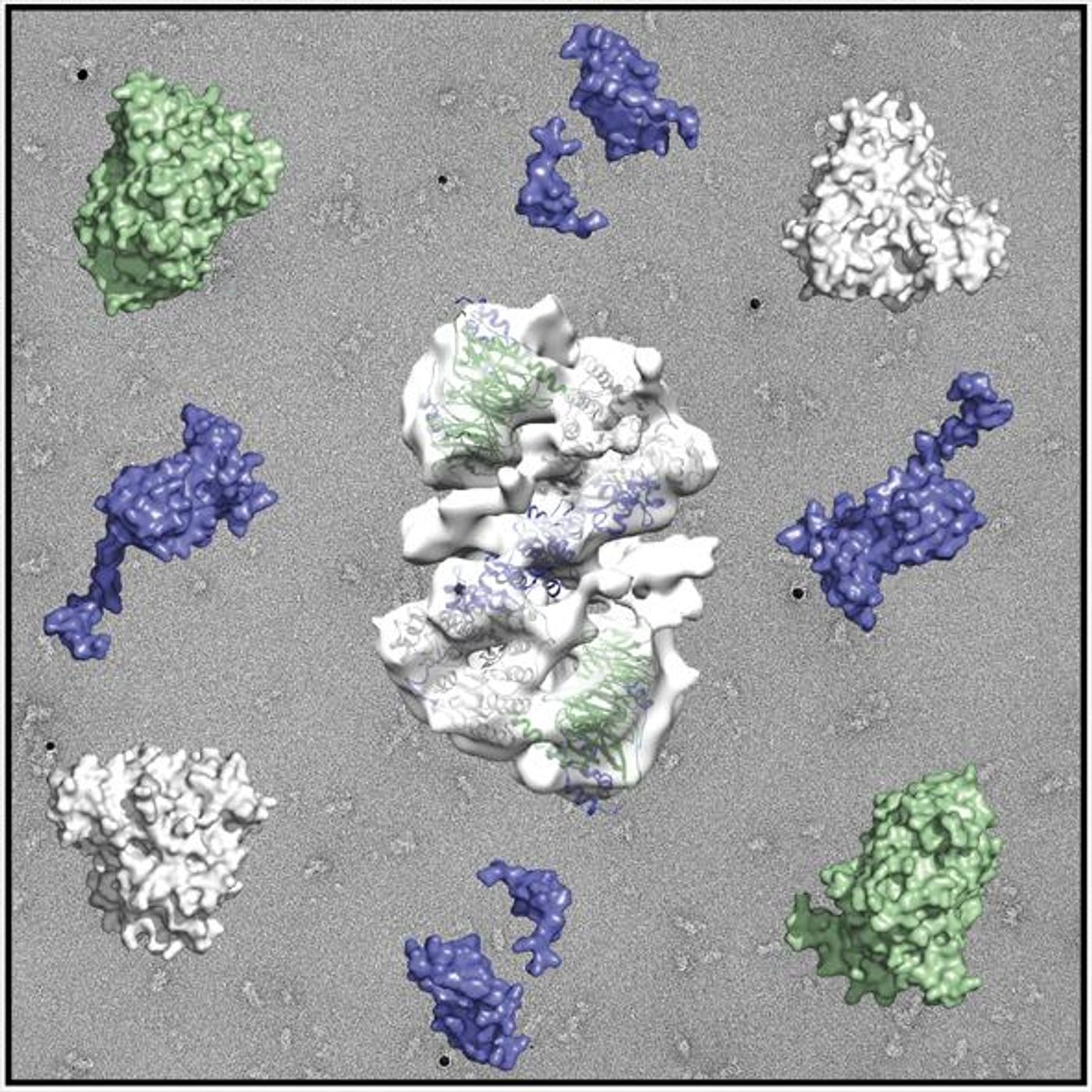
In hopes to both understand the three-dimensional structure of the NuRD complex proteins as well as characterize their molecular nature, University of Leicester researchers studied images using electron microscopy. Their results were recently published in the journal
eLIFE.
HDAC1 and MTA1 are known to exist at high levels in some incidences of cancer, and studying the interaction between these proteins as well as the relationship between MTA1 and RBBP4 in cancer patients and healthy patients could give the researchers a clue on how to protect disease-preventing equilibrium.
Electron microscopy showed an “extensive interface” between MTA1 and RBBP4. The three-dimensional images also showed a “repression complex” in cases where equilibrium was disrupted from NuRD complex-related activity. Specifically, the findings showed a second recruitment site within MTA1 for RBBP4, not known to exist before this study. Based on the different combinations of conformations between the three core proteins, the complexes are able to bind to different histones at different times. Understanding the components of this repression complex and the entire “architecture” of the NuRD complex will help scientists identify which mechanisms they should be targeting as a treatment option.
“An understanding [of] the exact mechanism by which the NuRD complex assembles and functions could make it an attractive target for developing novel and specific anti-cancer therapeutics and treatment for a range of genetic diseases,” said University of Leicester’s Dr. Christopher Millard.
Now that the University of Leicester research team has developed a more complete model of the NuRD complex, they can move on to understanding what goes wrong in NuRD signaling in cases of cancer or genetic diseases.
Sources:
University of Leister,
eLIFE