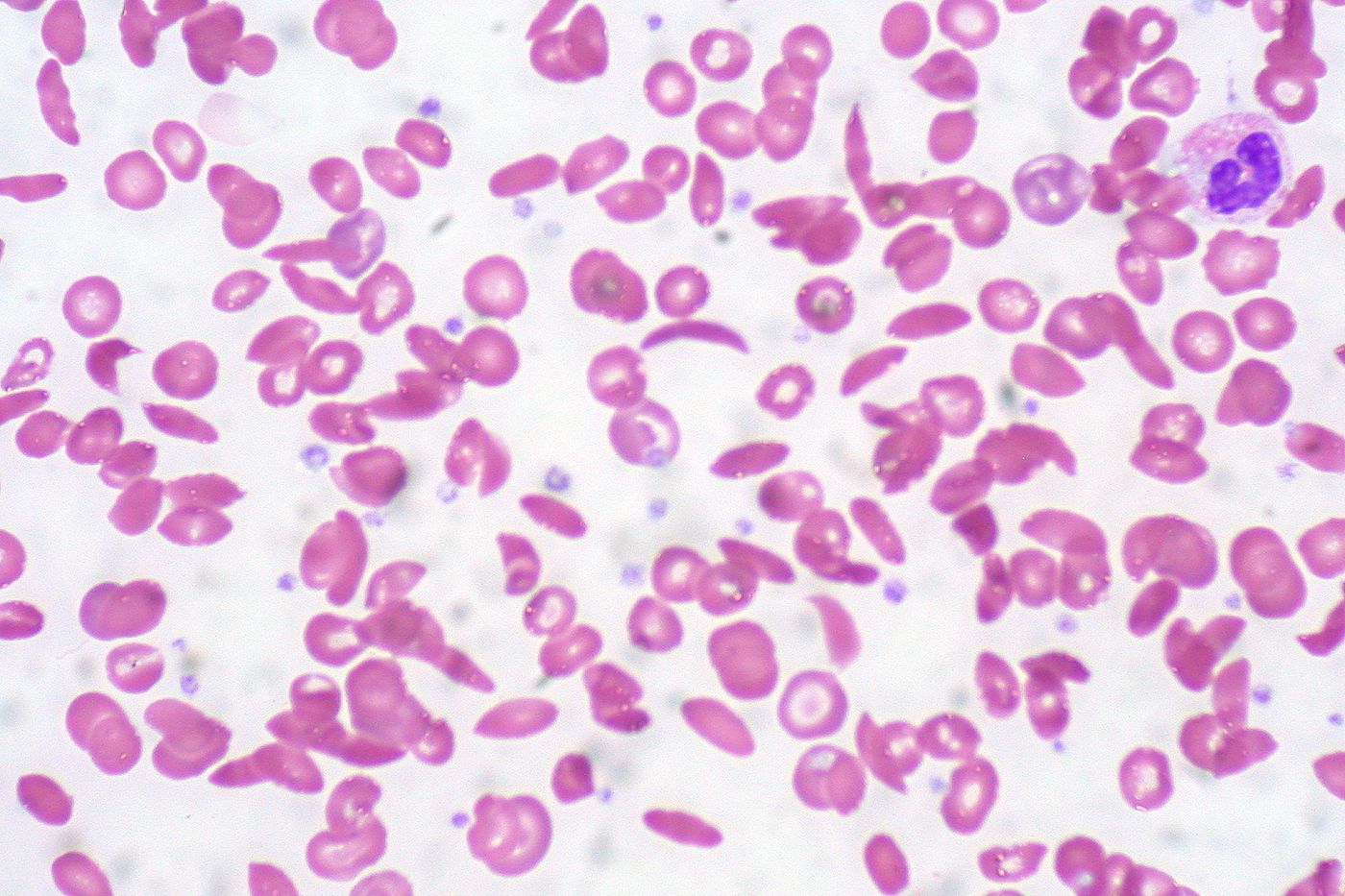
Cardiovascular problems are among the top causes of death in people with sickle cell anemia, a genetic disease caused by a mutation in the beta-globin gene. In a new study from the Cincinnati Children’s Hospital Medical Center, scientists made it their mission to get to the bottom of the heart complications that occur uniquely in patients with sickle cell anemia.
Using a mouse model of sickle cell anemia, Cincinnati researcher performed a series of non-invasive tests to investigate the effect sickle cell anemia has on the heart. Tests included cardiac MRI, electrocardiography, microscopy of isolated heart tissue cross sections, and a transcriptome analysis to understand the presence and level of gene expression of certain genes associated with heart function.
In order to be assured that the results from the tests were unique to cases of sickle cell anemia, researchers compared the findings to those of mice with chronic, iron-deficient anemia.
The unique results that they ended up with identified a so-called “restrictive cardiomyopathy” associated with an enlargement of the heart occurring because of sickle cell anemia. The diseased heart muscle “predisposes the myocardium to electrophysiological abnormalities and sudden death,” the authors write.
From the transcriptome analysis, the researchers found further confirmation of cardiovascular complications from sickle cell anemia: upregulation of genes causing increased oxidation, hypoxia and fibrosis of cardiovascular tissue, in addition to down regulation of genes vital for successful electrophysiological function.
The gene expression trends the researchers derived from the transcriptome analysis were all observed right before many mice models of sickle cell anemia died suddenly.
Studies continue on heart complications associated with sickle cell anemia; the sickled blood cells that characterize the disease clog blood vessels, putting people at a higher than normal risk for infection in addition to organ damage from oxygen deficiency. Using genetic knockout mice, scientists are still looking at identifying the specific molecular mechanisms and pathways that trigger cardiovascular fibrosis. “Identification of these pathways will allow development of new targeted therapies to treat cardiac dysfunction in people with sickle cell anemia,” said senior author Punam Malik, MD.
Clinical trials will soon be underway to test humans with the same non-invasive tests used successfully in the mouse model study. Whether induced by sickle cell anemia or not, detecting fibrosis in the heart as early as possible is vital to achieving the best prognosis possible for patients with cardiovascular complications.
This study was recently published in the journal
Proceedings of the National Academy of Sciences.
Sources:
Cincinnati Children’s Hospital Medical Center,
National Heart, Lung, and Blood Institute