Of all the different cell types circulating through the bloodstream, platelets are the cells we have to thank for instigating the clotting process when a blood vessel is damaged. Scientists are now starting to fully understand how platelets know when it’s time to begin clotting, and it’s all about the translation of mechanical forces into biochemical signals.
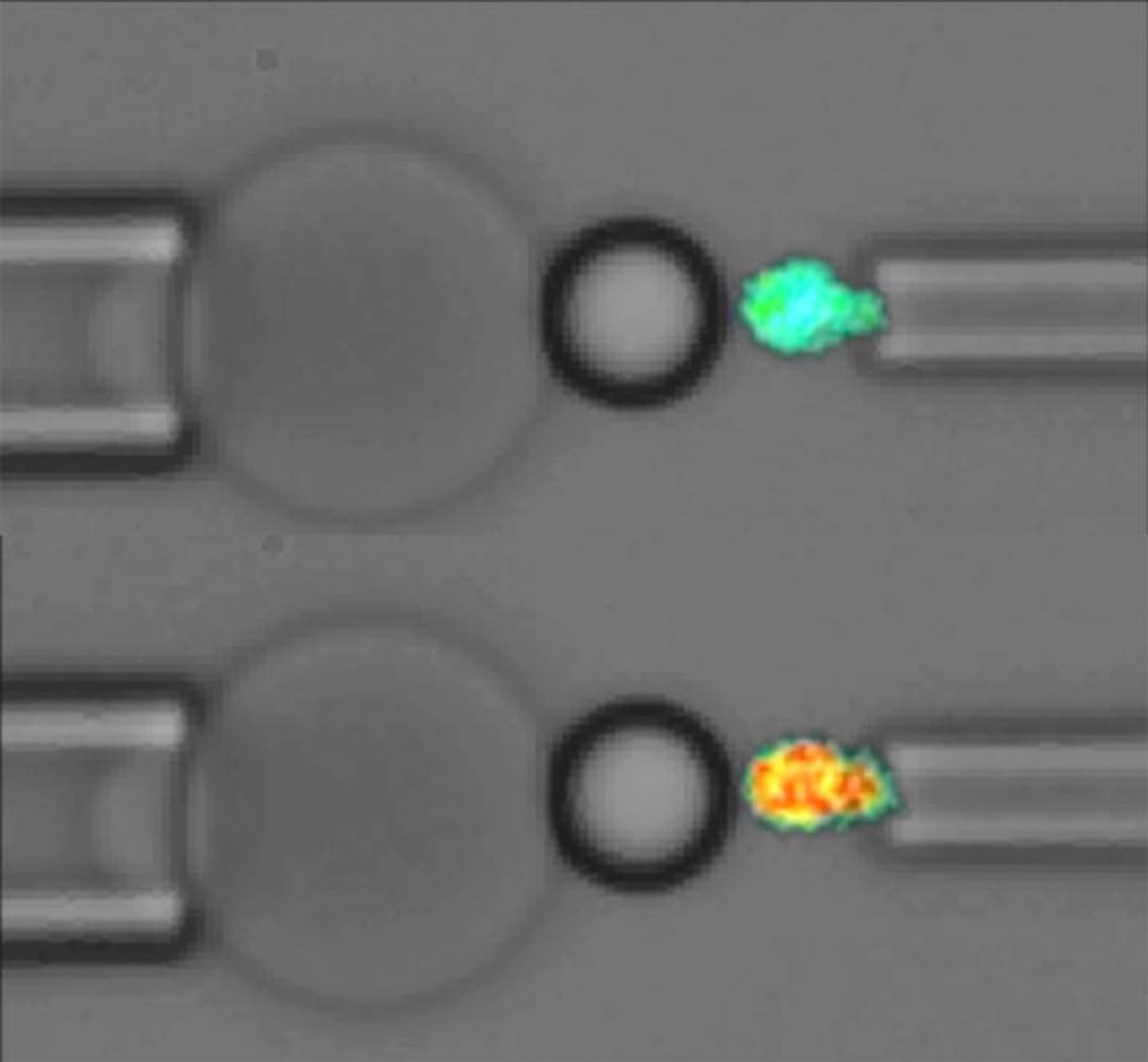
In a first-of-its-kind study, scientists from the Georgia Institute of Technology used a unique tool called a “fluorescence biomembrane force probe” to measure the force applied to a single molecule on a platelet. “We visualized the conformational changes in a single protein and the subsequent signaling event inside a cell at the same time," explained Lining (Arnold) Ju, PhD.
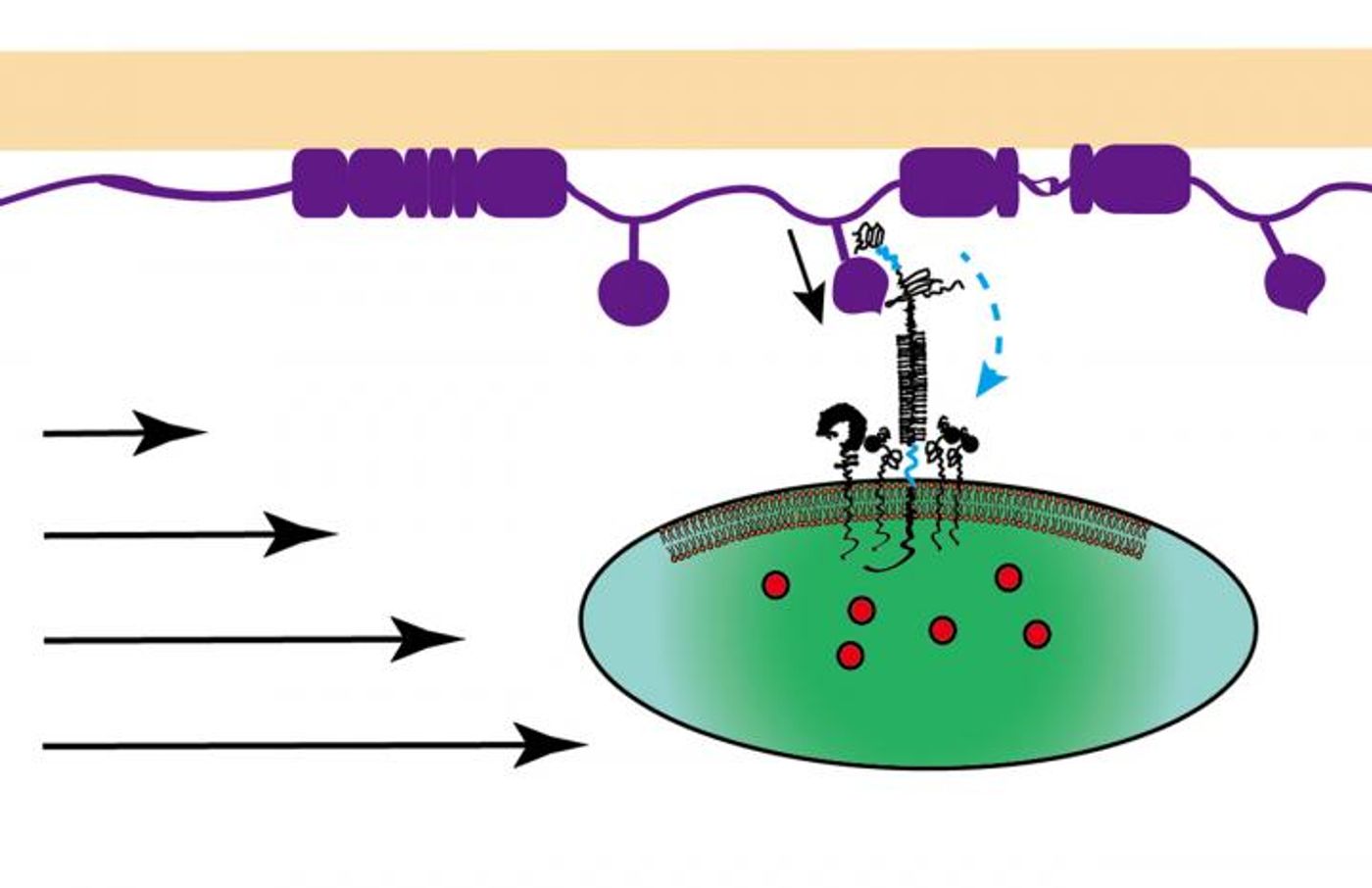
Ju and the rest of the team used the probe to apply mechanical forces to a protein called glycoprotein Iba (GPIba), which human platelets use to receive mechanical signals for transduction into chemical signals. The study, published in the journal
eLife, was the “first detailed mechanobiology study” displaying the process of GPIba on blood platelets transforming mechanical forces into chemical signals.
While applying mechanical forces to GPIba, researchers observed a couple of different reactions occur. One is the release of different calcium ions, which “alter adhesion between platelets and other components of the clotting process,” a sign of the conversion of mechanical information into chemical signals.
They also saw two geometrically separate domains of the GPIba protein unfold in a synergistic manner, an event that gives platelets detailed information about the magnitude and length of the mechanical forces acting upon it. Along with information gleaned about the mechanical forces from the release of calcium ions, platelets are then equipped with the tools to trigger the appropriate clotting process.
"These properties generate cooperativity - a synergistic effect that results in the highest signaling quantity and quality at an optimal force where it lasts the longest,” said Professor Yunfeng Chen, PhD.
Like many aspects of human physiology, both excessive clotting and inadequate clotting can cause complications in the body. A bleeding disorder called von Willebrand disease (VWD) occurs when a clotting protein, von Willebrand factor (VWF), is defective or absent completely. Individuals with VWD are prone to frequent nosebleeds, bruising, and hemorrhage after “invasive” situations like surgery, pregnancy, and blunt trauma.
Past studies have attributed VWD to a failure of cells to form a “platelet plug” on the walls of injured blood vessels, but Professor Cheng Zhu, another scientist involved in the project, said their research “reveals another defect.”
They found that the mutation of VWF associated with Type 2B VWD, which is believed to affect 15 to 30 percent of VWD patients, “abolishes the synergy” between the two distinct unfolding events of the GPIba protein. Without the synergy of these two events, the GPIba molecule is unable to effectively turn mechanical forces into biochemical signals to trigger clotting. “The mechano-sensing machinery doesn’t work well in the presence of this mutation,” Zhu explained. “The platelet just doesn’t get the signal that would activate it.”
The collaborative effort led by Zhu, Ju, Chen, and countless others lays the foundation for future treatments for VWD, either with genetic editing to mend the mutation and/or developing drugs to help GPIba successfully translate mechanical information into chemical information. Additionally, these findings highlight new targets for therapeutic intervention in cases of excessive clotting, a complication that is known to lead to myocardial infarction, stroke, and even death.
Lastly, this research also has the potential to expand to other cellular systems that similarly recruit proteins to translate mechanical forces into chemical information.
Sources:
Georgia Institute of Technology,
National Hemophilia Foundation,
Johns Hopkins Medicine Heart & Vascular Institute
Images: Lining (Arnold) Ju, John Toon, Yunfeng Chen