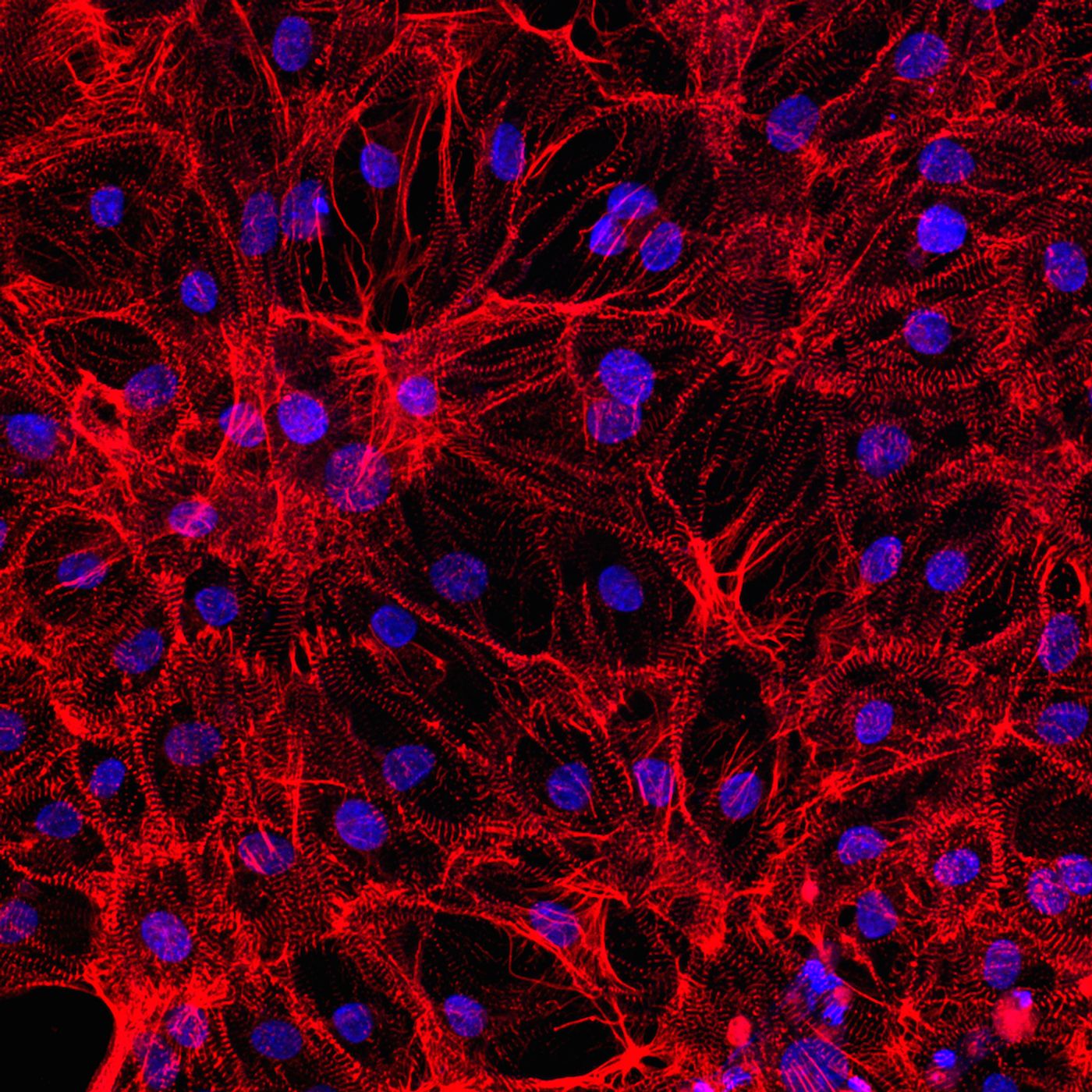
For unknown reasons, some people are sensitive to certain medications to treat heart conditions like cardiomyopathy. Scientists have discovered a new, safe way to predict which people are at risk for complications from these medications, using induced pluripotent stem cells (iPS cells).
"Thirty percent of drugs in clinical trials are eventually withdrawn due to safety concerns, which often involve adverse cardiac effects," said Joseph Wu, MD, PhD, director of Stanford University's Cardiovascular Institute. "This study shows that these cells serve as a functional readout to predict how a patient's heart might respond to particular drug treatments and identify those who should avoid certain treatments."
“Growing” heart muscle cells, called cardiomyocytes, from iPS cells essentially results in a carbon copy of an individual’s original cells, making them prime subjects for testing for cardiotoxicity. Plus, this idea could work for other cell types and other medical conditions as well.
Scientists from the Stanford University Medical Center studied the cardiomyocytes they constructed from iPS cells obtained from seven people, all without any known genetic predisposition for cardiac problems. Researchers sequenced the genetic material from the cardiomyocytes’ RNA to decipher the quality and amount of proteins made by these cells. After comparing their results among the seven study participants, the researchers had a near-complete idea of the gene expression patterns in cardiomyocytes.
“Gene expression patterns of the iPS cell-derived cardiomyocytes from each individual patient correlated very well,” said lead author of the study Elena Matsa, PhD. “But there was marked variability among the seven people, particularly in genes involved in metabolism and stress responses.”
With the normal patterns of expression in mind, the scientists then tested the cardiomyocyte reaction to two drugs: rosiglitazone, used to treat type 2 diabetes, and tacrolimus, an immunosuppressant used to prevent rejection of transplanted organs. From past studies and clinical trials, these drugs are known to cause adverse cardiac effects in some but not all people. Until now, scientists haven’t had a dependable method for predicting which people are not well-suited to receive these drugs, making their prescription quite a predicament.
The cardiomyocytes made out of the iPS cells from only one out of the seven participants showed “very abnormal expression of genes in a key metabolic pathway,” and this set of cardiomyocytes was also unique from the others in the response to rosiglitazone. These cardiomyocytes produced abnormal amounts of reactive oxygen species (ROS), byproducts of certain reactions that have the potential to cause “a number of deleterious events.” Additionally, in the presence of rosiglitazone, these cardiomyocytes were unable to regenerate mitochondria, which severely inhibited their ability to provide cellular energy, and contracted much more weakly than the cardiomyocytes from the other six study participants.
“I think the patient-derived iPS cell platform gives us a surrogate window into the body and allows us to not only predict the body's function but also to learn more about key disease-associated pathways."
The researchers could not pinpoint a specific gene mutation to be responsible for the abnormal cardiomyocyte response. However, they did identify an entire metabolic pathway that appeared to be behind the response to rosiglitazone. Plus, they were successful in fixing the problems in the pathway with genome editing, restoring normal function and a response to rosiglitazone that was more like that of the other participants’ cardiomyocytes.
“There are only a few examples of how to carry out [precision medicine] in a clinically meaningful way,” Wu said. “I think the patient-derived iPS cell platform gives us a surrogate window into the body and allows us to not only predict the body's function but also to learn more about key disease-associated pathways."
At the end of the study, Matsa and her team created more cardiomyocytes from iPS cells, this time from three different study subjects who had either undergone a heart biopsy or received a transplant. By comparing the cardiomyocytes derived from iPS cells to the native heart tissue of the three subjects, Matsa could confidently confirm that the gene expression patterns in the native cells and in the cells made in the lab were significantly similar.
Matsa’s study provides solid evidence that cardiomyocytes derived from human iPS cells have significantly similar genetic profiles to native host cardiomyocytes. Moreover, the study shows that these iPS cell-derived cardiomyocytes can be used to predict in the lab an individual’s response to certain cardiac drugs, which can pinpoint which individuals without any signs of abnormalities are in fact at risk for complications from certain medications.
The study was recently published in the journal
Cell Stem Cell.
Sources:
Stanford University Medical Center,
BioTek
Image: AxolBio