Who is MOST At Risk for Blood Clots in the Heart?
In the United States, heart disease is dangerously common and very expensive. Many people with heart disease and at a high risk of blood clots can benefit from preventative medication, but scientists are interested in identifying the most at-risk patients for prescribing these drugs to cut down on negative side effects.
Over several years, a collaborative effort from scientists at Ohio State University and Johns Hopkins University resulted in a new method for predicting these at-risk individuals, using a quantifier called “mitral jet penetration.”
Mitral jet penetration is a measure of how much blood successfully travels through the mitral valve into the left ventricle. If not enough blood makes it into the ventricle, the collection of blood in the heart’s chambers can lead to blood clots and strokes, making mitral jet penetration a critical factor in the development of blood clots.
In the study, researchers measured this critical factor with “E-wave propagation index” (EPI), which they then used to determine an individual’s risk of blood clots. "The beauty of the index is that it doesn't require any additional measurements. It simply reformulates echocardiogram data into a new metric," said computational fluid dynamics expert Rajat Mittal, one of the principal investigators of the study. "The clinician doesn't have to do any additional work."
Blood clots occur whenever blood is idle for a relatively long time, making the heart chambers the most at-risk areas for clot development because they are the “largest reservoirs of blood in the body.” Clots also often occur when the ejection fraction of blood from the left ventricle is reduced from a healthy level of 55 to 70 percent to an unhealthy, dangerous level of just 15 percent, but ejection fraction not necessarily an accurate predictor of future clotting risk.
"Because we understood the fluid dynamics in the heart using our computational models, we reached the conclusion that the ejection fraction is not a very accurate measure of flow stasis in the left ventricle," Mittal said. "We showed very clearly that the ejection fraction is not able to differentiate a large fraction of these patient and stratify risk, whereas this E-wave propagation index can very accurately stratify who will get a clot and who will not.”
Doctors can prescribe anticoagulants to prevent blood clots, but these drugs often have negative side effects. Predicting which patients are most at risk and prescribing only them with anticoagulant drugs might be the best approach.
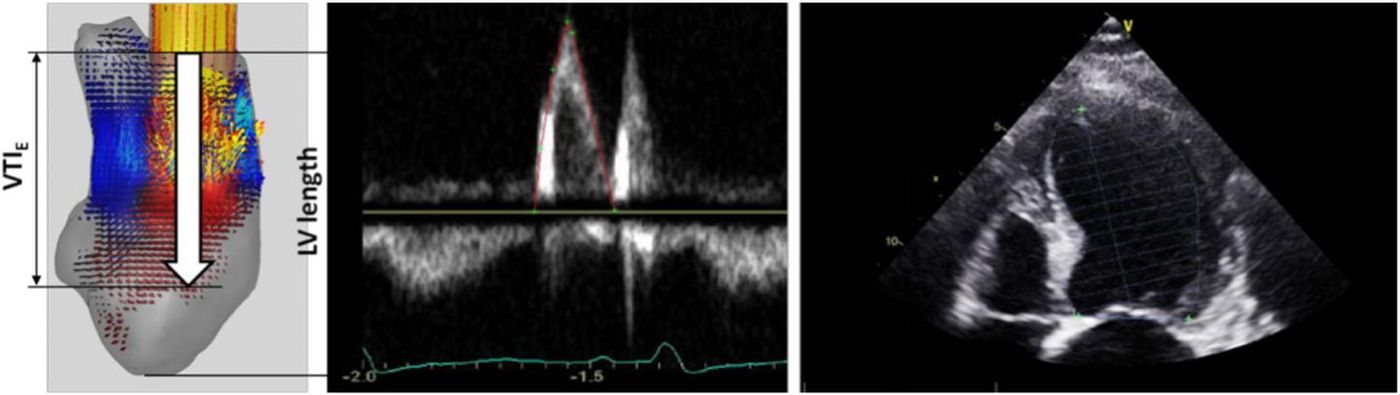
Image: The velocity time integral of the E-wave as measured from pulse Doppler at the mitral valve leaflet tips. Source: Rajat Mittal, Jung Hee Seo and Thura Harfi
The study would not have been possible without the “Stampede” supercomputer at the Texas Advanced Computing Center, where they completed simulations after designing their original hypothesis, based on specific data from 13 patients, including fluid flow, physical structures, and biochemistry.
In addition to the supercomputer simulations, which took several thousand computing hours to complete, they used data from 75 individuals used to test the hypothesis: 25 healthy patients, 25 patients with experienced blood clots in their left ventricle, 25 patients who had a “compromised heart” but no blood clots.
Combining the simulations and the patient analysis and looking at EPI numbers, researchers found that nearly one in every five patients with severe cardiomyopathy, meaning any disease of the heart muscle, is at risk of a left ventricular clot. If they are not already prescribed, researchers suggest anticoagulation medications for these patients.
"Computational fluid dynamics is such an established way of studying phenomena in mechanical engineering, but has rarely been tried in humans,” explained lead author Thura Harfi, from Ohio State University. “But now, with the development of high-resolution cardiac imaging techniques like cardiac computed tomography (CT) and the availability of supercomputing power, we can apply the power of computational fluid dynamics simulations to study blood flow in human beings.”
The findings from the new study can be applied to new diagnostic tools for clinicians and new, efficient computational models. Ronald Joslin from the National Science Foundation, which unded the study, said that a “basic understanding of the physics of how blood flows in the heart to real-time predictions and treatments for the well-being of patients.”
Harfi’s study was recently published in the International Journal of Cardiology.
Sources: University of Texas at Austin, Texas Advanced Computing Center, National Heart, Lung, and Blood Institute