Tracing Cell Lineages Back to the Beginning of the Heart's Chambers
The heart’s four chambers, made up of left and right atria and ventricles, work together in an amazingly intricate fashion, and understandably the path embryonic cells take to build these chambers is just as complex.
Researchers from the Mindich Child Health and Development Institute at the Icahn School of Medicine at Mount Sinai Hospital dove deeply into the study of one gene that impacts the development of one type of heart chamber but not the other, hoping to answer some questions about the risk of congenital heart defects, which often result from complications during chamber formation.
"An in-depth understanding of the formation of the heart chambers will enable us to better comprehend the biology behind detrimental heart defects and how best to address them," explained lead investigator of the Nature Communications study, Nicole Dubois, PhD.
Dubois and her team examined a mice model capable of exhibiting the entire lineage of cells that build the chambers of the heart. They focused on a protein-coding gene called Foxa2 that appeared to be heavily involved during embryonic development.
As a protein-coding gene, Foxa2 regulates the transcription of other genes involved in embryonic development, which means that Foxa2 has a say in whether or not certain genes are expressed and active. Development is a complex process where dozens of different tissue types must differentiate from progenitors, or precursors, cells which are destined to become a certain tissue but start out with essentially a blank slate.
They found that the progenitor cells expressing Foxa2 early in the development process eventually turn into ventricular cardiomyocytes, or heart cells, but not atrial cardiomyocytes. This means that the distinction between atria and ventricles is made long before structural differences are evident between the two chamber types.
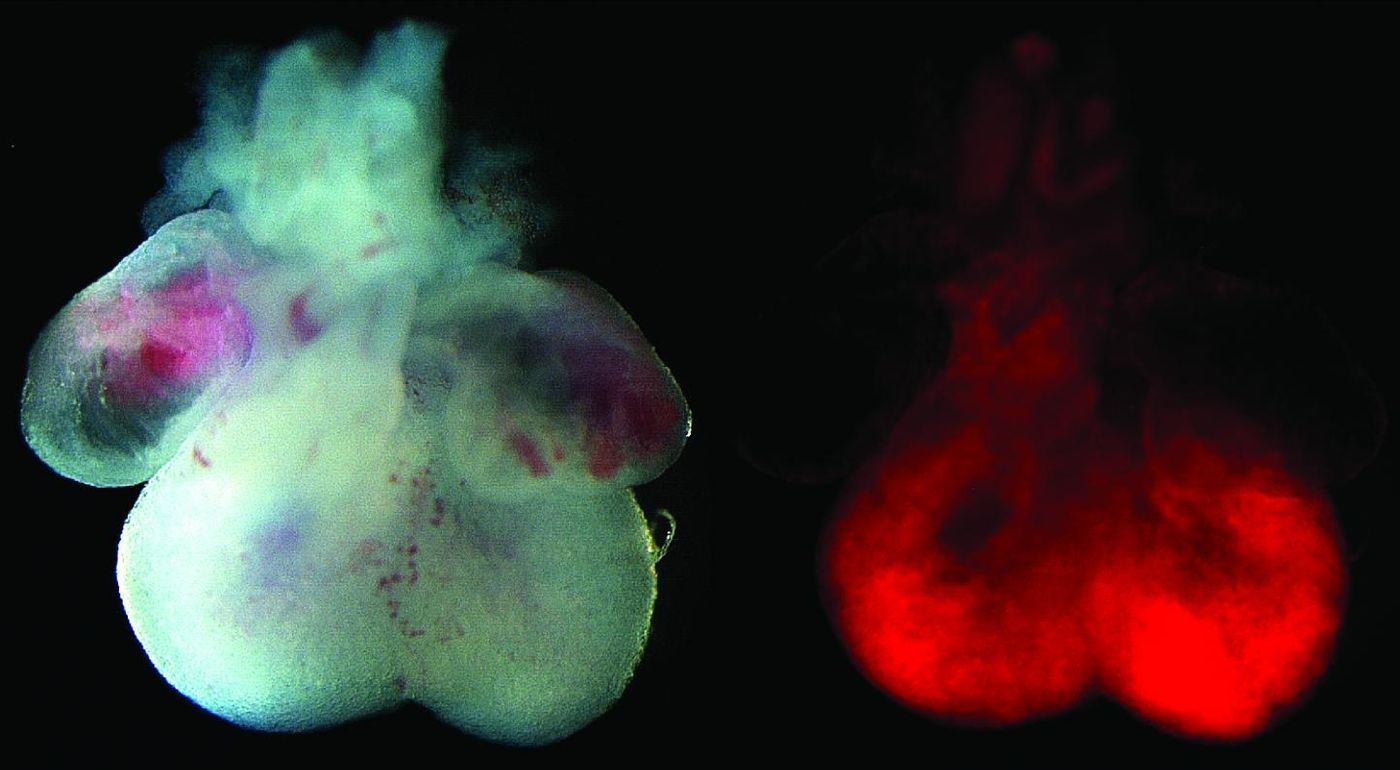
Image: The left image is of an embryonic mouse heart, showing the four chambered structure with atria at the top and ventricles at the bottom. The right image is the fluorescent lineage tracing reporter, showing that our newly discovered progenitor cell population contributes specifically to the ventricular chambers of the heart. Credit: Mount Sinai Health System
“We hope that these findings will also lead to new protocols for the generation of ventricular cardiomyocytes in cell culture that could potentially be used in therapeutic settings," Dubois said.
Dubois and first author Evan Bardot, a PhD student, still have many questions about the role of Foxa2 during embryonic heart development that have yet to be addressed. Whatever the answers may be, they could open an entirely new window into the understanding of congenital heart defects, the most common type of birth defect.
Sources: GeneCards Human Gene Database, Mount Sinai Hospital/School of Medicine