Excess Fat in the Heart Interferes with Energy Production
Energy production in heart cells, like all other cells, takes place in the mitochondria, the organelles famous for being the “powerhouse of the cell.” However, sometimes mitochondria use too much fat, a phenomenon often associated with diabetes and obesity. In a new study from the University of Iowa, researchers show how excess fat in the heart can interfere with cells’ ability to produce energy.
When heart cells use more fat than they need, the heart muscle’s ability to adapt to metabolic changes is impaired. Researchers from the new study believe that dysfunction in cardiac energy production is potentially responsible for the increased risk of heart failure experienced by virtually everyone with diabetes, a condition that impacts nearly 30 million Americans.
“One of the cardinal manifestations of the hearts of people with diabetes is the tendency to overuse fat as a metabolic fuel, which ultimately leads to mitochondrial and cardiac damage," explained study leader E. Dale Abel, MD, PhD.
The mitochondria normally use fatty acids as fuel, but if fatty acids aren’t available, then other molecules can be used, like glucose, lactate, and ketone bodies.
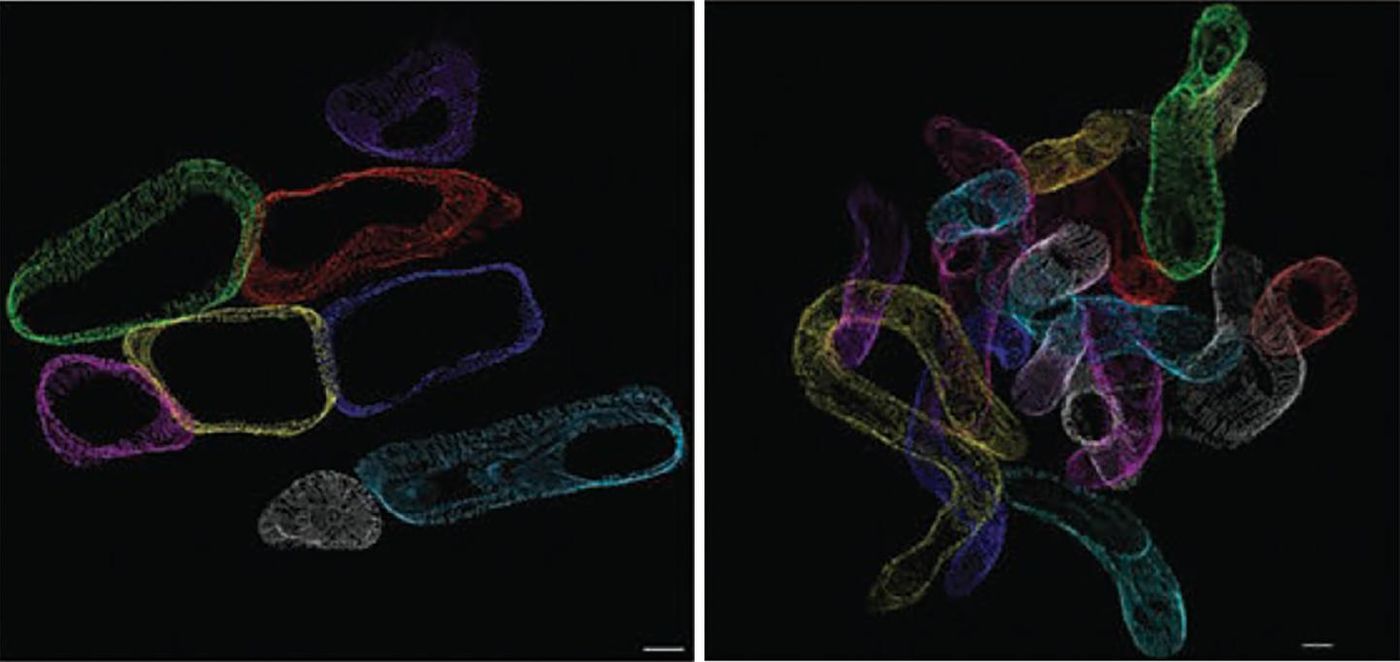
Abel’s study incorporated mice genetically modified so that their mitochondria metabolize excess amounts of fat, demonstrating what happens in the human heart in many individuals with diabetes. The research team used a three-dimensional electron microscopic cellular imaging technique so they could “directly observe structural changes to the mitochondria.” By doing so, they saw that these energy-producing organelles became thinner and “more twisted.”
These structural changes in mitochondria as a result of excess fat lead to less efficient energy production. And the more fat that heart cells consume as energy, the more drastic the changes cardiac mitochondria go through in structure and function.
The new study was also able to elucidate the connection between excess fat metabolism and mitochondrial changes: over a relatively long period of time, excess fat leads to a build up of reactive oxygen species (ROS), a group of reactive molecules that instigate cellular damage. Removing ROS reversed the effects of excess fat, returning mitochondria to their appropriate structure and function.
Interestingly enough, removing ROS in normal heart cells - with normally-sized mitochondria - resulted in mitochondria growing four-fold, suggesting, researchers explain, that ROS levels are “inversely proportional to mitochondria size.”
"We have demonstrated and detected how increasing the amount of fat (lipid) that the heart consumes leads to dramatic changes in the structure and function of the mitochondria in the heart,” Abel explained. “These studies provide a new window into how these changes to mitochondria could occur in the lipid-overloaded heart."
The present study was published in the journal Circulation Research.
Sources: BioTek, University of Iowa Health Care