Creating Stem Cells to See Heart Disease Like Never Before
A discovery arose from growing human arterial endothelial cells in the lab, and scientists from the Morgridge Institute for Research couldn’t be more thrilled about the future possibilities. A new method that allowed them to grow such cells provides a new opportunity to apply their findings to developing drugs that could treat arteriosclerosis.
Arteriosclerosis is a general term for when the arteries become hardened, and it is often also referred to as atherosclerosis. Hardened arteries increase the risk of heart disease because blood has a more difficult time traversing the arteries to reach the body’s tissues, which need oxygen and nutrients carried in the blood.
The new study, published in the journal Stem Cell Reports, builds on previous research from the same group in the summer of 2017. In this study, scientists described the first-ever production of functional arterial cells from pluripotent stem cells “at both the quality and scale to be relevant for tissue engineering.”
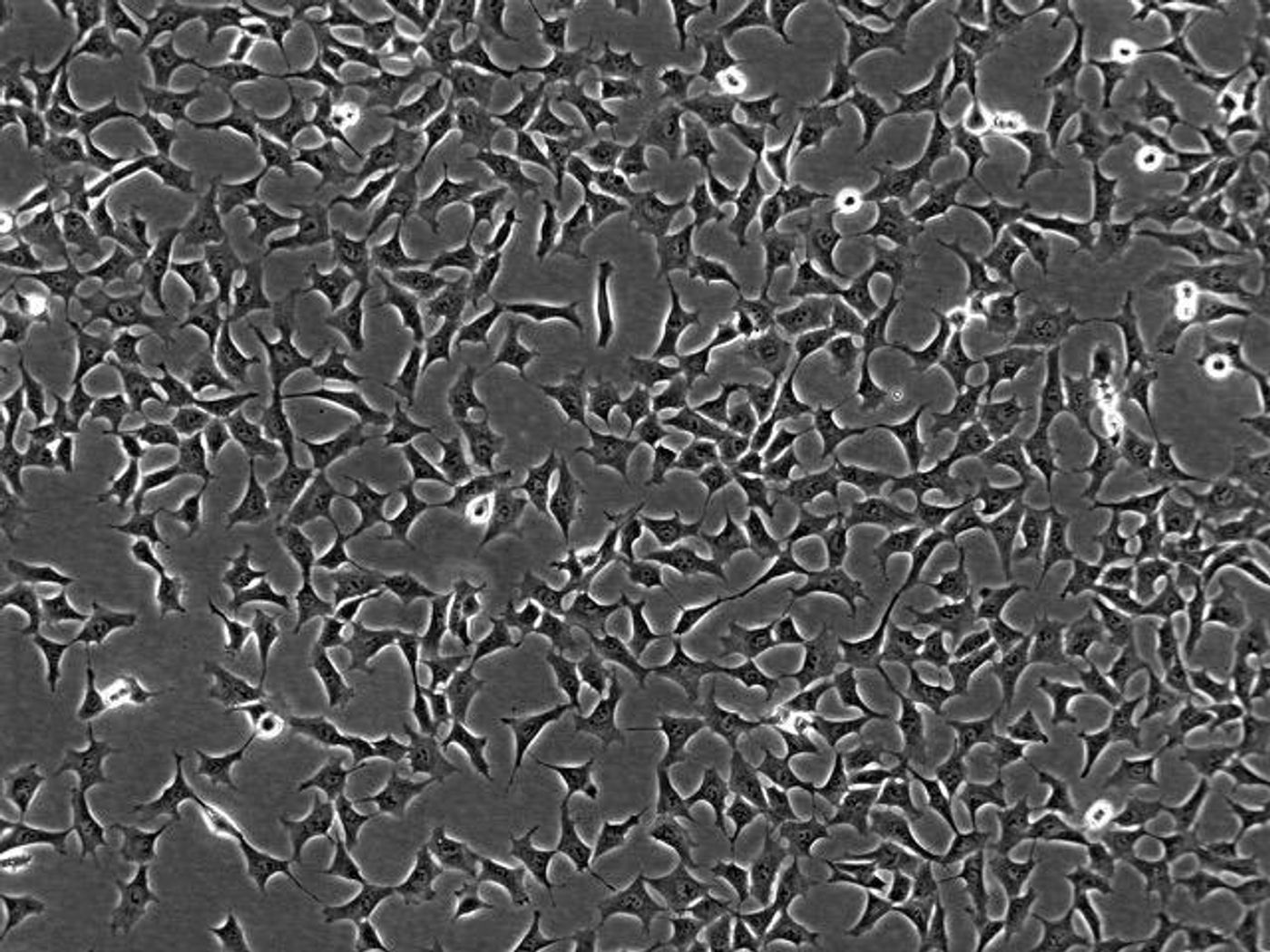
In the new study, researchers used two transcription factors that come together uniquely when combined to “almost exclusively” regulate arterial endothelial cell growth. Via cord blood and adult bone marrow tissue, the researchers were able to develop a new method of growing human arterial endothelial cells in the lab. Historically, these cells are infamous for being extremely difficult to grow.
After successfully growing stable amounts of the endothelial cells, researchers found that they grew in two specific forms, one that resisted the transition to mesenchymal cells and one that did not. The so-called “endothelial to mesenchymal transition” is a known risk factor for heart disease, linked to hardening of the arteries.
The harmful potential of mesenchymal cells stems from their structure: star-shaped cells that tend to clump together. On the other hand, endothelial cells are elongated and smooth, forming single-cell layers. But what makes one type of arterial endothelial cells resist the endothelial/mesenchymal transition?
"My suspicion is there is a naturally occurring transition that's probably healthy, like in a response to injury," explained study leader Dave Vereide. "This is probably part of the normal homeostatic balance. What I think happens during disease progression is that the balance shifts, and now there's too much transition taking place and you get these massive deleterious effects."
Identifying two types of endothelial cells is just the beginning. Going forward, Vereide and his team plan on investigating further to pinpoint genetic differences between cell types that makes one more resistant than the other to transitioning into mesenchymal cells. Afterward, they could potentially develop drugs based on these genetic differences to prevent endothelial cells from turning into mesenchymal cells, thus avoiding damage to the blood vessels in the context of heart disease.
Vereide also suggests a clinical test to “predict whether your cells are going to be permissive or resistant to the transition and steer clinicians to the cells that will perform best in the human body.”
Sources: Morgridge Institute for Research, Mayo Clinic